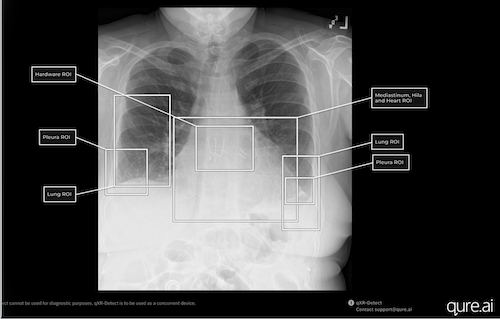
Qure.ai qXR-Detect provides bounding boxes and indicates the location of suspicious regions of interest. Photo: Qure.ai
Feb. 26, 2026 — The U.S. Food and Drug Administration (FDA) has given 510(k) class II clearance of qXR-Detect, the latest computer-assisted detection (CADe) radiography solution from Qure.ai. It powers the early identification, highlighting and categorizing of key positive findings on plain film chest X-rays to support emergency room (ER) physicians, family medicine practitioners and radiologists.
Chest radiography is the most performed imaging exam today in the US, with 70 million obtained each year in the ER, outpatient department or part of routine health assessments. This "6 in 1" indication FDA clearance, completes the entire qXR product suite of AI algorithms, covering the breadth of findings encountered on chest X-rays. Findings that can be categorized into six regions of interest are added including — lung; pleura; mediastinum /hila & heart; bone; hardware and others.
“There are more than 100 potential findings on a plain film chest X-ray that AI tools can help detect and assist radiologists with, supporting earlier recognition of abnormalities,” states Professor Amit Gupta, Division Chief of Cardiothoracic Imaging and Modality Director of Diagnostic Radiography at University Hospitals Cleveland Medical Center. “Tools such as qXR-Detect go beyond binary detection alone. In addition to flagging potential abnormalities, the system provides visual localization and explainability, such as through bounding boxes and region-of-interest labels, which helps the interpreting radiologist quickly understand where and why an alert has been generated. When used alongside standard clinical interpretation, chest radiography augmented with AI-based detection and localization can offer a practical frontline support tool for identifying findings such as lung nodules and other abnormalities that may warrant closer attention or further diagnostic workup.”
“These super six indications cleared by the FDA for qXR-Detect establishes Qure.ai as the foremost commercial AI company, with the most comprehensive regulatory clearances for “qXR-Detect is also the only chest X-ray CADe device cleared by the FDA with a Predetermined Change Control Plan (PCCP). This will help assure U.S. health system customers access to the most up-to-date version of the algorithm as models and architecture evolves. This new clearance is testimony to the robust FDA regulatory process Qure.ai upholds and an exciting moment for all involved in the quest to create a stage shift in lung cancer.”
To achieve the FDA clearance, a performance test of qXR-Detect was conducted as a standalone and a multi-reader multi-case (MRMC) clinical study. The device showed good performance in both and met the predefined success criteria.
Qure.ai’s FDA cleared indications now total 26 across 9 products for X-ray and CT, exceeding 65 CE certified indications and other global validations. The latest FDA clearance adds breadth to the product portfolio in the US with the existing clearances for identification and quantification of lung nodules; triage and notification of pneumothorax and pleural effusion; plus, multiple neurocritical finding clearances including for intracranial hemorrhage, cranial fracture and midline shift.
Go to www.qure.ai for more information.
* Regulatory clearances of AI algorithms vary by geographic region
Related information on Qure.ai


 April 20, 2026
April 20, 2026 









