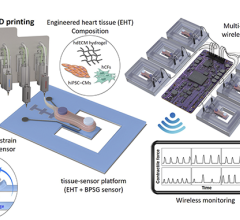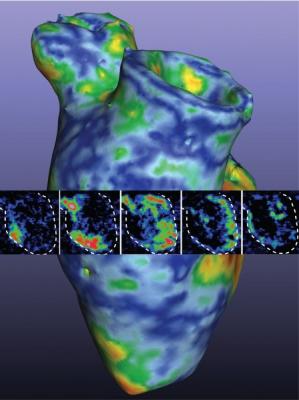
This image was used on the cover of the American Heart Association’s Circulation Research journal is a 3-D rendering of the printed heart pump developed at the University of Minnesota. The discovery could have major implications for studying heart disease. Kupfer, Lin, et al., University of Minnesota.
July 16, 2020 — In a groundbreaking new study, researchers at the University of Minnesota have 3-D printed a functioning centimeter-scale human heart pump in the lab. The discovery could have major implications for studying heart disease, the leading cause of death in the United States killing more than 600,000 people a year.
The study is published and appears on the cover of Circulation Research, a publication of the American Heart Association (AHA).[1]
In the past, researchers have tried to 3-D print cardiomyocytes, or heart muscle cells, that were derived from what are called pluripotent human stem cells. Pluripotent stem cells are cells with the potential to develop into any type of cell in the body. Researchers would reprogram these stem cells to heart muscle cells and then use specialized 3-D printers to print them within a three-dimensional structure, called an extracellular matrix. The problem was that scientists could never reach critical cell density for the heart muscle cells to actually function.
In this new study, University of Minnesota researchers flipped the process, and it worked.
“At first, we tried 3-D printing cardiomyocytes, and we failed, too,” said Brenda Ogle, the lead researcher on the study and head of the Department of Biomedical Engineering in the University of Minnesota College of Science and Engineering. “So with our team’s expertise in stem cell research and 3-D printing, we decided to try a new approach. We optimized the specialized ink made from extracellular matrix proteins, combined the ink with human stem cells and used the ink-plus-cells to 3-D print the chambered structure. The stem cells were expanded to high cell densities in the structure first, and then we differentiated them to the heart muscle cells.”
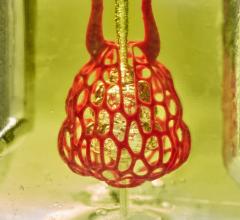
What the team found was that for the first time ever they could achieve the goal of high cell density within less than a month to allow the cells to beat together, just like a human heart.
“After years of research, we were ready to give up and then two of my biomedical engineering Ph.D. students, Molly Kupfer and Wei-Han Lin, suggested we try printing the stem cells first,” said Ogle, who also serves as director of the University of Minnesota’s Stem Cell Institute. “We decided to give it one last try. I couldn’t believe it when we looked at the dish in the lab and saw the whole thing contracting spontaneously and synchronously and able to move fluid.”
Ogle said this is also a critical advance in heart research because this new study shows how they were able to 3-D print heart muscle cells in a way that the cells could organize and work together. Because the cells were differentiating right next to each other it was more similar to how the stem cells would grow in the body and then undergo specification to heart muscle cells.
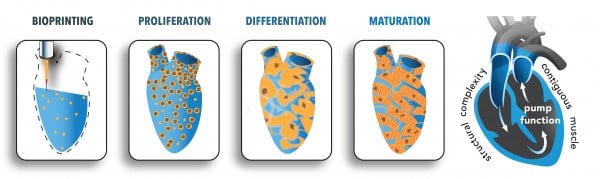
This schematic illustrates the steps involved in generating the human heart pump.
Compared to other high-profile research in the past, Ogle said this discovery creates a structure that is like a closed sac with a fluid inlet and fluid outlet, where they can measure how a heart moves blood within the body. This makes it an invaluable tool for studying heart function.
“We now have a model to track and trace what is happening at the cell and molecular level in pump structure that begins to approximate the human heart,” Ogle said. “We can introduce disease and damage into the model and then study the effects of medicines and other therapeutics.”
The heart muscle model is about 1.5 centimeters long and was specifically designed to fit into the abdominal cavity of a mouse for further study.
“All of this seems like a simple concept, but how you achieve this is quite complex. We see the potential and think that our new discovery could have a transformative effect on heart research,“ Ogle said.
This research was primarily funded by the National Institutes of Health (National Heart Lung and Blood Institute, National Institute of Biomedical Imaging and Bioengineering, and National Institute of General Medical Science) with additional funding from the National Science Foundation Graduate Research Fellowship Project and the University of Minnesota Doctoral Dissertation Fellowship.
To read the full research paper entitled “In Situ Expansion, Differentiation and Electromechanical Coupling of Human Cardiac Muscle in a 3D Bioprinted, Chambered Organoid.”
In addition to Ogle, Kupfer and Lin, other University of Minnesota researchers involved include University of Minnesota College of Science and Engineering faculty Professor Alena G. Tolkacheva (biomedical engineering) and Professor Michael McAlpine (mechanical engineering); University of Minnesota Medical School Associate Professor DeWayne Townsend (integrative biology and physiology); current and former University of Minnesota master’s, Ph.D. students and postdocs Vasanth Ravikumar (electrical engineering), Kaiyan Qiu (Ph.D., mechanical engineering), and Didarul B. Bhuiyan (Ph.D.), Megan Lenz (M.S.), and Ryan R. Mahutga (biomedical engineering); and undergraduate student Jeffrey Ai (biomedical engineering). The team also included University of Alabama Department of Biomedical Engineering Professor and Chair Jianyi Zhang and University of Alabama biomedical engineering Ph.D. student Lu Wang and research associate Ling Gao (Ph.D.).
Another recent development in July 2020 is a team in Australia that is 3-D printing skin for burn victims. Read more in the article Robot That Prints New Skin Could Revolutionize Burns Treatment.
Related 3-D printing Content:
The Future of 3-D Printing in Medicine
World’s Smallest Intravascular OCT Imaging Device Created Using 3-D Printing
New Technique Allows More Complicated 3-D Bioprinting
The Use of 3-D Printing in Cardiology
Reference:

 October 21, 2024
October 21, 2024