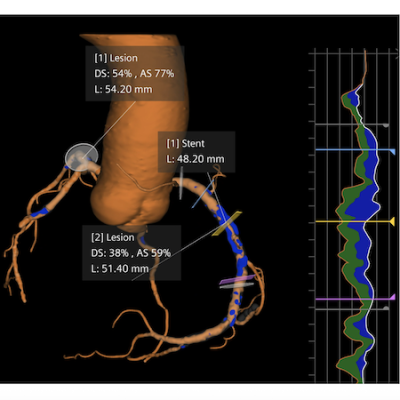
Detailed anatomical and morphological information support more precise plaque assessment and can inform key intraprocedural decisions, such as lesion preparation, stent length and landing-zone selection. (Photo: Siemens Healthineers)
Dec. 2, 2025 — At RSNA 2025, Siemens Healthineers announced Syngo.CT Coronary Cockpit1, a new software solution within the Syngo.via2 platform designed to support the management of coronary artery disease (CAD) for both dual-source and photon-counting CT scanners. As the leading cause of death globally and a major contributor to disability,3 CAD continues to pose a significant global health challenge. This drives increasing demand for diagnostic and treatment planning tools that are both efficient and minimally invasive. Syngo.CT Coronary Cockpit addresses these needs by providing advanced, automated plaque analysis. Critical details embedded in cardiac CT images thus become accessible, supporting clinical decision-making and intervention planning.
Dr. Balint Szilveszter, Heart and Vascular Centre, Semmelweis University, Budapest, Hungary, commented on the clinical value of the new solution: “There is a lot of information that is locked within CT images. Syngo.CT Coronary Cockpit is an automated tool that provides a fast approach to unlock this information to include it in our everyday clinical routine. The information can be used for optimizing medical therapy or to plan PCI procedures before even entering the cath lab.”
Traditionally, patients with suspected or known CAD are often referred to the cath lab for diagnostic procedures, even though many of these could be avoided. Advances in cardiac CT, particularly with dual-source and photon-counting CT scanners, have made non-invasive coronary CT angiography (CCTA) a frontline diagnostic tool. Studies have shown that using CCTA with the Naeotom Alpha CT scanner from Siemens Healthineers — combining photon-counting technology with dual-source — can more than halve referrals for diagnostic angiography, lower costs, and shorten hospital stays4, thanks to its diagnostic value in assessing plaque visibility.
To further enhance diagnostic confidence, Syngo.CT Coronary Cockpit characterizes both the amount and the morphology of coronary plaque through AI-based segmentation, labeling, and visualization. Information such as plaque composition, size, and distribution supports clinicians in better assessing risk and selecting the most appropriate treatment, whether medical management or intervention.
Integration of Syngo.CT Coronary Cockpit with dual-source and photon-counting CT scanners of Siemens Healthineers enables high native temporal resolution and supports clinical accuracy and operational efficiency across the CAD pathway. Having detailed anatomical and morphological information available before an interventional procedure may enhance the efficiency of the cath lab and optimize resource utilization. By providing such insights in the cath lab, clinicians gain an overview of coronary anatomy and pathology. This additional insight supports more precise plaque assessment and can inform key intraprocedural decisions, such as lesion preparation, stent length, and landing-zone selection. Ultimately, this enables more patients to receive personalized care. The use of non-invasive CCTA supported by advanced analysis tools means faster, more precise diagnosis and treatment planning, with the potential to avoid unnecessary invasive procedures and their associated risks.
“With Syngo.CT Coronary Cockpit, we combine our expertise in CT imaging and interventional therapy to help people with coronary artery disease live longer, healthier lives,” said Philipp Fischer, head of Computed Tomography at Siemens Healthineers. “This joint approach paves the way for a future where CT not only diagnoses but actively guides treatment decisions. It marks an important step toward CT-guided PCI and brings us closer to personalized, minimally invasive care.”
- Syngo.CT Coronary Cockpit is pending 510(k) clearance and is not yet commercially available in the United States.
- Syngo.via can be used as a standalone device or together with a variety of Syngo.via-based software options, which are medical devices in their own right. Syngo.via and the Syngo.via based software options are not commercially available in all countries. Due to regulatory reasons its future availability cannot be guaranteed. Please contact your local Siemens Healthineers organization for further details.
- JACC: Global prevalence of coronary artery disease: an update from the global burden of disease study
- https://doi.org/10.1148/radiol.231234, https://doi.org/10.1148/ryct.230022


 October 08, 2025
October 08, 2025