October 29, 2010 – Site enrollment has been completed for the 50-site CITADEL and CENTURION clinical studies to evaluate the Tyrx AigisRx Antibacterial Envelope. The device is designed to help reduce surgical-site infections (SSIs) associated with cardiac implantable electronic devices (CIEDs).
The trials are both large, prospective, multicenter clinical studies, which together, will enroll 4,300 subjects at 50 clinical study sites across the United States. Patients enrolled in these studies have a pacemaker, implantable cardioverter-defibrillator (ICD) or a cardiac resynchronization therapy (CRT) device that will be replaced with an ICD or CRT device accompanied by AigisRx.
In the CITADEL study, prospectively enrolled patients receiving AigisRx will be compared to published controls who have undergone pacemaker, ICD or CRT device replacement with an ICD without AigisRx.
In the CENTURION study, prospectively enrolled patients will be compared to 2000 case-matched controls that have undergone pacemaker, ICD or CRT device replacement with a CRT without AigisRx.
The primary endpoints for both studies will be 1) major ICD/CRT device-related infections and 2) ICD/CRT device-related mechanical complications. Patients will be followed for 12 months, with predefined interim analyses at three and six months.
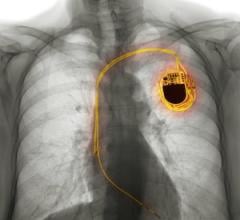
The AigisRx Envelope is an antibacterial mesh envelope designed to deliver antimicrobial agents which help provide protection against infections associated with implanted pacemakers and ICDs/CRTs. AigisRx also securely holds a pacemaker or ICD/CRT in order to create a stable environment when implanted in the body.
“Published data shows us that the risk of CIED infection is higher for ICDs/CRTs in comparison to pacemakers. Additionally, replacement procedures carry more infection risk compared to initial implants. The CITADEL and CENTURION studies will provide vital information on the clinical performance of AIGISRx in these particularly high risk groups for CIED infection,” said Scott Burkett, M.D., FACP, FACC, Burkett Heart Clinic, Monroe, La.
Tyrx commercializes innovative, implantable combination drug/device products focused on infection control, including the AigisRx Antibacterial Envelope and AigisRx Flat Sheet products. Their products contain antimicrobial agents, rifampin and minocycline, which have been shown to reduce infection by organisms representing a majority of the infections reported in cardiac implantable electronic device (CIED)-related endocarditis, including “superbugs” or MRSA.
For more information: www.tyrx.com

 April 24, 2026
April 24, 2026 









