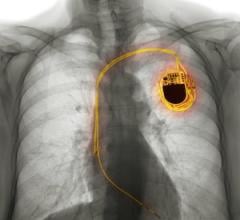
April 8, 2013 — Tyrx Inc. has received a license from Health Canada to market its new AigisRx R Fully Resorbable Antibacterial Envelope technology with implantable electronic devices (IEDs). The AigisRx R Antibacterial Envelope is specifically designed to stabilize IEDs while also releasing antimicrobial agents to help provide protection from microbial colonization of the device during surgical implantation. Tyrx is the leader in the commercialization of implantable medical devices intended to help reduce surgical site infections.
“Canada will be the first market in the world to gain access to our next generation AigisRx Antibacterial Envelope technology,” commented Robert White, Tyrx president and chief executive officer. “The AigisRx R technology includes all of the advantages of the original AigisRx, such as stability and infection reduction, but now with the added benefit of being fully resorbable.”
The AigisRx R Envelope is intended to hold the IED devices, such as pacemakers and implantable cardioverter defibrillators (ICDs) securely in place in order to provide a stable environment when implanted in the body. Additionally, the AigisRx R contains the antimicrobial agents rifampin and minocycline, which are released over a seven to 10 day period, in order to help provide protection from microbial colonization of the device during surgical implantation.
“Due to the continued increase in surgical site infections associated with cardiac implantable electronic devices (CIEDs), we are very pleased to have access to the AigisRx R Antibacterial Envelope. We hope that AigisRx R will serve as a key technology in addressing the significant impact that surgical site infections have on patient morbidity, mortality, and health care system costs,” stated Peter Guerra, head of electrophysiology at the Montreal Heart Institute.
Tyrx Inc. commercializes innovative, implantable combination drug+device products focused on infection control, including the AigisRx Antibacterial Envelope, designed to reduce surgical site infections associated with CIEDs. AigisRx products contain the antimicrobial agents, rifampin and minocycline, which have been shown to reduce infection by pathogens responsible for the majority of CIED infections, including “superbugs” such as methicillin-resistant S. aureus (MRSA).
For more information: www.TYRX.com, www.HeartDeviceInfection.com

 April 24, 2026
April 24, 2026 









