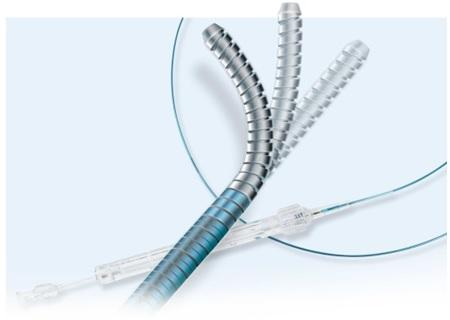
June 22, 2017 — The U.S. Food and Drug Administration (FDA) has identified Vascular Solutions’ recent recall of its Venture catheters as a Class I recall, it announced this week. This is the most serious type of recall, because the FDA said use of these devices may cause serious injuries or death.
Vascular Solutions initiated a voluntary recall of the guidewires April 25, 2017. The company said there is a risk of the catheter tip splitting or separating during use. Excess material at the tip of the catheter may separate and could embolize into the patient's bloodstream. This can result in serious adverse health consequences such as the development of blood clots, embolism of the excess material to vital organs, or death.
Recall is for Venture RX Catheter (Model 5820), Venture OTW (Model 5821) and Venture CS Catheter (Model 5822). These devices were distribution between May 7, 2015 to April 19, 2017, and manufactured between May 7, 2015 to March 31, 2017. The recall includes 7,054 nationwide in the U.S.
The Venture catheter is intended for directing, steering, controlling and supporting a guidewire to access veins and arteries in the arms, legs, hands, feet, and heart muscle (myocardium). Certain models may also administer saline fluids or drugs into blood vessels.
The company sent an urgent medical device recall letter to all affected customers. The letter asked distributors and customers to identify and remove any affected Venture catheters from inventory and quarantine. This includes completing a field safety notice and a customer inventory form. After all the affected catheters are returned, customers are asked to complete the Vascular Solutions Inc. Distributor Inventory Form, and return it via email to [email protected]. Upon receipt of the form, Vascular Solutions' customer service department will provide a return authorization number and arrange for return of affected Venture catheters. A credit will be offered after affected devices have been returned, the company said.
Healthcare professionals and consumers may contact Vascular Solutions at (888) 240-6001 Monday through Friday, between the hours of 8 a.m. and 5 p.m. Central Time, or by e-mail at [email protected] with any questions related to this recall.
For a complete list of recalled product codes and lot numbers go to www.fda.gov/MedicalDevices/Safety/ListofRecalls/ucm564092.htm?source=govdelivery&utm_medium=email&utm_source=govdelivery
Related Content:
VIDEO: Basics of Interventional Guidewire Design and Function
Understanding the Design and Function of Guidewire Technology
The Basics of Guide Wire Technology


 May 13, 2026
May 13, 2026 









