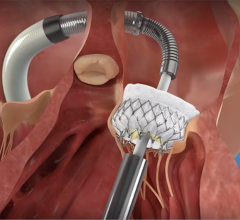
August 17, 2012 —Xeridiem, a vertically integrated OEM that designs, develops and manufactures complex single-use medical devices, announced it will provide LoneStar Heart Inc. with design and development services for the minimally invasive delivery system for Algisyl-LVR, an implantable biopolymer for the treatment of dilated cardiomyopathy.
LoneStar Heart is developing a trio of technologies with the potential to restore the heart wall structure and prevent or reverse the progression of advanced heart failure. "This development collaboration will allow LoneStar Heart to focus on their core expertise, which is further developing their Algisyl-LVR technology while relying on Xeridiem for the engineering and design control infrastructure. Essentially Xeridiem will be acting as LoneStar Heart's development team," said Joseph Lee, president of Xeridiem.
Xeridiem designs, develops and manufactures single-use medical devices focused in gastroenterology, cardiology and urology for established healthcare companies and venture-backed entrepreneurs. Since 1986, Xeridiem, as a silent partner, has enabled major medical device companies and venture-backed entrepreneurs to deliver more than 22 million medical devices used worldwide.
LoneStar Heart Inc. is developing cardiac restorative therapies for patients with heart failure that harness the heart's ability to repair itself. Based on its integrated cardiomechanical and biomolecular technologies, the privately held company is advancing a broad portfolio of products to restore the failing heart's structure and function in collaboration with the Texas Heart Institute, the University of Texas Southwestern Medical Center and a global network of leading clinicians. These products include Algisyl-LVR, cardiac stem-cell modulators, and cellular and genetic therapies delivered as standalone treatments, or in combination with the company's proprietary biopolymer matrix system.
For more information: www.xeridiem.com


 July 08, 2024
July 08, 2024