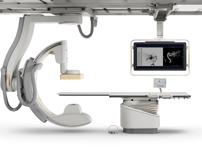
December 4, 2009 – In an effort to lower clinician radiation exposure in the cath lab, a new device displays the real-time dose each clinician receives and alerts them when they enter the radiation field. The DoseAware system was introduced by Philips Healthcare at the 2009 Radiological Society of North America (RSNA) annual meeting this week in Chicago.
DoseAware delivers real-time visualization, display and tracking of clinical staff X-ray dose to help healthcare professionals manage their exposure on the spot. Staff members wear a radiation detecting badge and their real-time detection level is shown on a screen attached to the cath lab monitors. When a clinician enters the radiation field, the screen warns the clinicians so they can take precautions to avoid exposure.
The system data is stored and can be used to generate reports on each staff member’s exposure and trending information on particular cath lab rooms.
The clinical tool is part of the Allura Xper angiography imaging system.
For more information: www.medical.philips.com


 April 01, 2026
April 01, 2026