July 11, 2019 — Mednax Inc. and Mednax Radiology Solutions announced that Chief Medical Officer Ricardo C. Cury, M.D., FSCCT, will present at the 14th Annual Scientific Meeting of the Society of Cardiovascular Computed Tomography (SCCT), July 11-14 in Baltimore. His session is titled “Building a National Centers of Excellence in Coronary Computed Tomography Angiography (CCTA) to Support Implementation in Clinical Practice.”
“Many emergency departments lack standardized CCTA care processes or have outdated ones. This results in significant patient care variations, which ultimately leads to longer lengths of stay that decrease patient satisfaction and increase costs,” said Cury. “The Mednax Radiology Solutions Cardiac Centers of Excellence Program and Baptist Health South Florida in Miami led the way to develop new CCTA protocols, subspecialist interpretation coverage, and training programs for emergency department physicians, hospitalists, cardiologists, nursing staff and technologists in appropriate CCTA utilization. As a result, Baptist Health realized dramatic gains in efficiency, cost savings and patient satisfaction, including a decrease in the average per-case episode cost by more than $1,000.”
In addition to the decrease in episode cost, Baptist Health’s new CCTA protocols lead to a:
-
Fifty (50) percent decrease in length of stay;
-
Low rate for major adverse cardiac events (MACE) with a negative predictive value of 99.8 percent; and
-
Significant (58.3 percent) decrease in effective radiation dose using iterative reconstructions and tailored protocols.
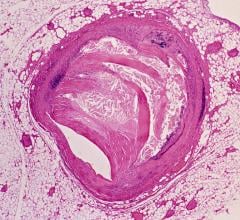
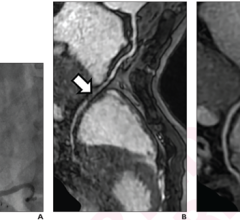
CCTA is a contrast-enhanced radiographic assessment of the coronary arteries. It is a non-invasive alternative that can show narrowed or blocked blood vessels. Although CCTA has been endorsed by several guidelines from the American College of Cardiology/American Heart Association, CCTA has been slow to be adopted for low- to intermediate-risk chest pain patients. Through its Centers of Excellence, Mednax works to leverage such best-practice programs across its national network and expand services and technology nationwide to provide better and faster care for patients.
Watch the VIDEO: Trends In Cardiac CT Imaging at SCCT 2015, an interview with Cury
For more information: www.mednax.com/radiology

 October 24, 2025
October 24, 2025