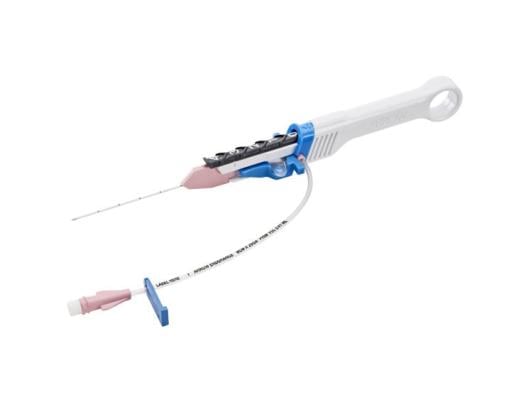
June 26, 2023 — The U.S. Food and Drug Administration (FDA) has issued a Class I recall on the Teleflex and Arrow International ARROW Endurance Extended Dwell Peripheral Catheter System after reports of catheter separation or leakage. If the catheter separates while in a blood vessel, the catheter fragments could be left in the bloodstream, and may migrate to other places in the body. This issue may cause serious injury including blockage of blood vessels, inadequate blood flow, injury to blood vessel walls, blood clots, blockage of the lung arteries (pulmonary embolism), heart attack, or death.
The FDA has identified this as a Class I recall, the most serious type of recall. Use of these devices may cause serious injuries, serious health consequences, or death.
Recalled Product
- Product Name: ARROW Endurance Extended Dwell Peripheral Catheter System
- Product Codes: See Recall Database Entry
- Devices Recalled in the U.S.: 262,016
- Dates Distributed: October 26, 2018 to May 10, 2023
- Date Initiated by Firm: May 19, 2023
Device Use
The ARROW Endurance Extended Dwell Peripheral Catheter System allows health care providers access to a patient’s peripheral vascular system, for short term use, to sample blood, monitor blood pressure, or administer fluids, blood, and blood products.
Reason for Recall
Teleflex, and their subsidiary Arrow International, are recalling the ARROW Endurance Extended Dwell Peripheral Catheter System after reports of catheter separation or leakage. If the catheter separates while in a blood vessel, the catheter fragments could be left in the bloodstream and may migrate to other places in the body.
This issue may cause serious injury, including blockage of blood vessels, inadequate blood flow, injury to blood vessel walls, blood clots, blockage of the lung arteries (pulmonary embolism), heart attack, or death.
Teleflex/Arrow International reports 83 complaints related to this issue. There have been 18 reported injuries and no deaths.
Who May Be Affected
- People who receive care with the ARROW Endurance Extended Dwell Peripheral Catheter System.
What to Do
On May 19, 2023, Teleflex and Arrow International sent an Urgent Medical Device Recall letter to customers that included the following recommended actions:
For Medical Facilities
- Immediately:
- Check inventory for product within the scope of this recall.
- Stop using and distributing affected product.
- Quarantine affected product.
- Mark the applicable checkbox (affected product or no affected product) on the acknowledgement form included with the letter. Fax the form to 1-855-419-8507 to Attn: Customer Service or email the form to [email protected].
- If you have affected product, a customer service representative will contact you with a Return Goods Authorization (RGA) number and will provide instructions for the return of affected products to Teleflex. Teleflex (or the local dealer) will issue a credit note upon receipt of the returned affected product.
- Ensure you only list batch numbers in scope of the recall notice when completing the form.
For Distributors
- Provide a copy of the Urgent Medical Device Recall letter to all customers who have received impacted product. Each customer must complete the acknowledgement form and return it to the distributor.
- Immediately stop use and distribution of, and immediately quarantine, the affected product. Then return all product in scope.
- Confirm to Teleflex that field activities outlined have been completed.
- After completing actions, forward the completed acknowledgement form to [email protected].
- Ensure you only list batch numbers in scope of the recall notice when completing the form.
- If product was distributed outside of the country, notify Teleflex Customer Service.
Contact Information
Customers with questions should contact Teleflex and Arrow International Customer Service:
Phone: 1-866-396-2111
FAX: 1-855-419-8507
Email: [email protected]


 May 13, 2026
May 13, 2026 









