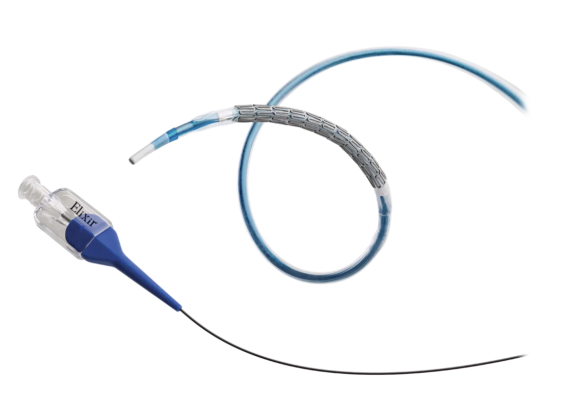
October 10, 2023 — Elixir Medical, a developer of innovative cardiovascular technologies, announced it will present primary endpoint and 6-month data from its DESyne BDS Plus Randomized Clinical Trial (RCT), a prospective, multicenter, single-blind study, as part of The Innovation Program at the Transcatheter Cardiovascular Therapeutics (TCT) conference in San Francisco. The data compare DESyne BDS Plus, the world’s first triple drug-eluting stent with local anticoagulation delivery technology using two anticoagulants (rivaroxaban and argatroban) and an anti-proliferative mTOR inhibitor (sirolimus) to a second-generation durable polymer DES.
“Stent thrombosis after percutaneous coronary intervention (PCI), though rare, is a severe complication with death or heart attack risk of up to 50% after the event. Dual antiplatelet therapy (DAPT) has played an integral role in keeping stent thrombosis rates low, but requires patient compliance for a minimum of six months and is associated with an increased risk of major bleeding,” said Motasim Sirhan, CEO of Elixir Medical. “DESyne BDS Plus with local anticoagulation delivery technology was designed to help mitigate the variable of drug compliance and to simplify the tradeoff decision of balancing the ischemic and bleeding risk. We are pleased to share the exciting findings from the first trial of DESyne BDS Plus as our team continues to innovate in the PCI space with our multiple platforms of disruptive technologies.”
The DESyne BDS Plus RCT includes 202 patients across 14 sites in Europe, New Zealand and Brazil. An imaging subset of 60 patients had angiographic and optical coherence tomography (OCT) assessment completed at six months. Data collection will continue through three years.
The data will be presented in the session “TCT Innovation V: Innovation in Coronary Disease Diagnosis and Therapies” on October 25th from 8-9:30 AM PDT in the Innovation & Clinical Science Theater, Hall B at the Moscone Center.
Elixir Medical is also hosting the scientific symposium, “Innovation for PCI: Improved outcomes require disruption” featuring a panel of speakers including Robert W. Yeh, M.D., Mirvat Alasnag, M.D, Ignacio Cruz-Gonzalez, M.D., Ph.D., Roxana Mehran, M.D. and Stefan Verheye, M.D., Ph.D.
The Elixir team will also exhibit at booth 115.
Full list of presentation details:
Innovation session: TCT Innovation V: Innovation in Coronary Disease Diagnosis and Therapies
Title: A Randomized Clinical Trial of a Novel Triple Drug Eluting Coronary Implant Eluting Two Anticoagulants and Sirolimus: Six-Month Clinical and Imaging Results of the DESyne BDS Plus Study
Presenter: Stefan Verheye, M.D., Ph.D.
Date: Wednesday, October 25, 2023
Time: 8:00 AM - 9:30 AM PDT
Location: Innovation & Clinical Science Theater, Hall B
Scientific symposium
Title: Innovation for PCI: Improved Outcomes Require Disruption
Presenters: Robert W. Yeh, M.D., Mirvat Alasnag, M.D., Ignacio Cruz-Gonzalez, M.D., Ph.D., Roxana Mehran, M.D. and Stefan Verheye, M.D., Ph.D.
Date: Tuesday, October 24, 2023
Time: 12:30 PM PDT
Location: Presentation Theater 3, Room 215, Level 2
Spotlight session: Coronary Spotlight 5: Disruptive Stent Technologies
Title: DynamX Coronary Bioadaptor Technology
Presenter: David Erlinge, M.D., Ph.D.
Date: Tuesday, October 24, 2023
Time: 3:30 PM - 4:55 PM PDT
Location: Coronary Spotlight Theater, Hall E, Exhibition Level
Spotlight session: Coronary Spotlight 5: Disruptive Stent Technologies
Title: DESyne BDS Plus Stent
Presenter: Stefan Verheye, M.D., Ph.D.
Date: Tuesday, October 24, 2023
Time: 3:30 PM - 4:55 PM PDT
Location: Coronary Spotlight Theater, Hall E, Exhibition Level
Session: Vulnerable Plaque Studies
Title: Plaque Stabilization and Regression: Another Significant Benefit of DynamX Bioadaptor
Presenter: Stefan Verheye, M.D., Ph.D.
Date: Wednesday, October 25, 2023
Time: 10:00 AM - 10:54 AM PDT
Location: Moderated Abstracts Station 8 – Emerging Clinical Science & Research Sessions, Hall C, Exhibition Level
Session: BVS Studies II
Title: 12-Month Clinical Outcomes of Patients Treated with the Novel DynamX Bioadaptor: DynamX Hong Kong Registry
Presenter: Wai Kin Chi, M.D.
Date: October 25, 2023
Time: 11:00 AM - 12:03 PM PDT
Location: Moderated Abstracts Station 6 – Emerging Clinical Science & Research, Hall C, Exhibition Level
Session: BVS Studies II
Title: Positive Adaptive Remodeling Effect of the DynamX Bioadaptor in BIOADAPTOR RCT
Presenter: Mark Webster, M.D.
Date: October 25, 2023
Time: 11:00 AM - 12:03 PM PDT
Location: Moderated Abstracts Station 6 – Emerging Clinical Science & Research, Hall C, Exhibition Level
For more information: www.elixirmedical.com
Find more TCT23 conference coverage here
Related TCT Coverage:
CRF Announces TCT 2023 Late-breaking Clinical Trials
VIDEO: Precision Cardiovascular Medicine: The Next Frontier in Patient Care and Innovation
VIDEO: 3-Year Follow-up of the SYMPLICITY HTN-3 Trial
VIDEO: Unique Features of Coronary Artery Disease in Women
Cardiovascular Research Foundation’s (CRF) TCT 2022 in Review
Photo Gallery Highlighting TCT 2022
Cardiovascular Research Foundation’s (CRF) TCT 2022 in Review
TCT 2022 Honors Recipient of Thomas J. Linnemeier Young Investigator Award
TCT 2022 Announces Winner of Shark Tank Innovation Award
Educational Leaders Join Forces on Educational Opportunities
Top TCT 2022 Award Recipients announced by Cardiovascular Research Foundation (CRF)


 October 31, 2025
October 31, 2025 









