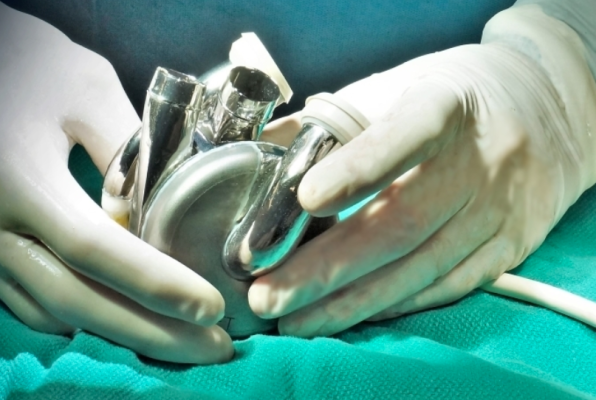
May 30, 2025 — BiVACOR, a clinical-stage medical device company developing the world’s first titanium Total Artificial Heart (TAH), announced that its device has received Breakthrough Device Designation from the U.S. Food and Drug Administration (FDA).
The designation supports the BiVACOR TAH as a bridge to transplant (BTT) for adults with severe biventricular or univentricular heart failure where current treatments, including LVADs, are not viable. The FDA’s Breakthrough Device program is reserved for technologies that may significantly improve outcomes for patients with life-threatening or irreversibly debilitating conditions. It offers priority regulatory interaction and accelerated pathways to approval.
“This is more than a regulatory milestone. It’s a validation of a concept we’ve spent decades proving that a fully implantable, total artificial heart isn’t just possible, it’s necessary,” said Daniel Timms, PhD, Founder and Chief Technology Officer of BiVACOR. “Patients with biventricular failure have been overlooked for too long. The early results from our clinical trial show that we can give them a second chance, without the compromises of older technologies. The Breakthrough Device Designation puts us on a faster track to deliver exactly that.”
The milestone follows the first phase of BiVACOR’s FDA Early Feasibility Study, where five patients in the U.S. received the TAH between July and November 2024. Based on positive safety and performance data, the FDA approved the expansion of the trial to include 15 additional patients starting later this year.
BiVACOR’s device represents a new category in artificial heart technology. Compact enough to fit most men and women, the TAH uses magnetic levitation, similar to maglev trains, to suspend a single dual-sided rotor. This rotor simultaneously powers the right and left circulatory systems, mimicking the natural heartbeat without valves or mechanical wear points. Its simplified design allows for pulsatile flow, large blood gaps to reduce trauma, and long-term durability.
“We’ve seen every kind of artificial heart technology over the last four decades, but this is the first system I’ve encountered that combines engineering elegance, efficiency, and safety with true clinical viability,” said William Cohn, MD, BiVACOR Chief Medical Officer and heart surgeon at the Texas Heart Institute. “The early results are remarkable with no strokes, no device-related complications, and a safety profile unlike anything in this space. With Breakthrough status in hand, we’re entering the next phase with the wind at our backs and real momentum to bring this to more patients.”
Heart failure affects more than 6 million Americans, and thousands of patients each year progress to irreversible biventricular failure. However, the number of available donor hearts remains stagnant, with fewer than 4,500 transplants performed annually in the U.S. BiVACOR is targeting this critical gap with a durable artificial replacement engineered for eventual long-term support.
The BiVACOR TAH is currently investigational and not approved for commercial use.
Learn more at www.bivacor.com.


 April 20, 2026
April 20, 2026 









