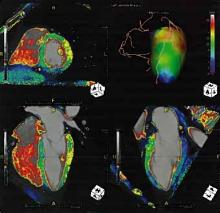
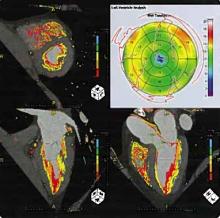
December 5, 2011 – New study shows significant increase in quality of life (QoL) among overweight patients following ablation of atrial fibrillation (AF), compared to patients with a lower body mass index (BMI) where no substantial improvement was observed. The study, published in the December edition of HeartRhythm, the official journal of the Heart Rhythm Society, reveals an association between BMI and a change in QoL between the pre- and postablation periods in a large AF patient cohort. The study also verifies comparable procedural success rates of ablation in both overweight and lean patients.