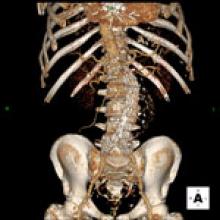
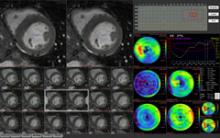
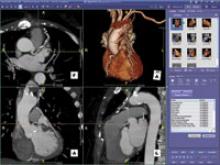
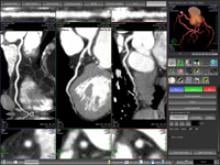
Our profession faces one of the most significant crises in its history,” said Michael Graham, M.D., president of the Society of Nuclear Medicine (SNM), regarding the shortage of nuclear medicine radioisotopes.
If you enjoy this content, please share it with a colleague
- Read more about Cardiac PET Adoption Gets a Boost
- Log in or register to post comments