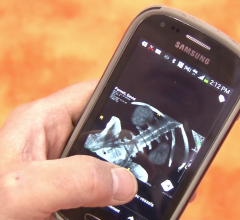
July 27, 2012 — Healthcare imaging specialist Barco announces the release of a new version of its MediCal QAWeb Mobile software for viewing digital imaging and communications in medicine (DICOM)-quality medical images on the iPad. The new release features a more intuitive user interface and is compatible with Apple’s third-generation iPad.
Designed to ensure fast visual calibration on the iPad, MediCal QAWeb Mobile offers a Visual Confidence Test featuring up to 20 dynamic test patterns. The application thus allows medical professionals to review – and even diagnose – clinical images on their Apple iPad when they are away from their workstation.
The new release also provides extra QA test methods to ensure confident viewing of clinical images on all three generations of iPad. Furthermore, in addition to stand-alone and embedded integration, the software now features a third and server-based integration option so radiologists can view DICOM-quality images using a simple web browser, without the need for a mobile image viewer.
Barco’s master file for its patented QAWeb Mobile methodology has been accepted by the U.S. Food and Drug Administration (FDA), which allows healthcare developers and integrators to apply for FDA 510(k) clearance when integrating Barco’s QA and calibration tool for mobile devices. Referencing the FDA master file speeds development and reduces the regulatory effort for FDA clearance.
A free trial version of Barco’s MediCal QAWeb Mobile software is available for download in the app store (itunes.apple.com) and can be used on iPad 1, iPad 2, and the new iPad.
For more information: www.barco.com/en/markets/healthcare

 April 05, 2019
April 05, 2019