CardiacAssist Launches Protek17 Arterial Cannula for TandemHeart pVAD
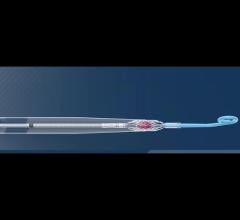
September 19, 2013 — CardiacAssist launched the Protek17 Arterial Cannula, a key component of the TandemHeart temporary circulatory support platform used to rest the heart and circulate blood for patients with severe cardiac dysfunction. Protek17 is designed for improved patient safety and ease of use, with new features including suture wings for secure attachment to the patient and a rubber stop to prevent over-insertion, a common cause of vascular access site bleeding.
The Protek17 cannula is U.S. Food and Drug Administration (FDA)-cleared and has been successfully deployed in multiple TandemHeart cases during a limited commercial release in 2013, and it is now available for full commercial distribution.
The FDA-cleared TandemHeart system has provided critical heart support to thousands of patients in the top cardiac centers worldwide. The system's left atrial cannulation creates a fully percutaneous extracorporeal blood circuit to bypass the left ventricle, augmenting the flow of blood by up to 5.0 L/min.
For more information: www.tandemheart.com, www.cardiacassist.com


 October 23, 2023
October 23, 2023