April 2, 2010 – Integrating a sleep disorder diagnostic tool into hospital cardiac care, the MARS Virtual Sleep Lab (VSL) was recently introduced at ACC 2010. It is the first device to provide a streamlined view of quantitative cardiac and sleep apnea analysis from any GE-monitored inpatient bed, helping enhance speed of diagnosis.
The system represents an important advance, as obstructive sleep apnea (OSA) impacts up to 50 percent of all heart disease patients.(1) When sleep disorders are diagnosed and treated early, it has been shown to improve cardiac conditions. Left undiagnosed, sleep apnea can increase heart disease risks.
More than 80 percent of individuals with moderate-to-severe OSA have not been clinically diagnosed.(2) Powered by the WideMed Ltd. Morpheus Hx sleep apnea diagnosis program, the system enables OSA diagnosis right from the hospital bed. This transforms in-patient rooms into virtual sleep labs, and enables a bedside diagnosis by improving utilization of monitored data to provide diagnostic information. According to a recent Journal of Clinical Sleep Medicine study, connecting the WideMed automated sleep analysis system to hospital monitors achieves results highly correlated with the standard test conducted in sleep labs – polysomnography.(3)
MARS VSL automatically reports on clinical data that is critical for making a sleep apnea diagnosis, such as apnea hypopnea index, sleep staging and respiratory events. Following a sleep apnea diagnosis, an AutoPAP device can be prescribed while the patient is still in the hospital. This increases the chance that OSA patients can begin treatment before other complications develop.
Holter Monitoring From Any Inpatient Bed
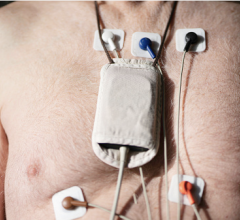
MARS VSL enables not only sleep apnea diagnostic testing, but also quantitative Holter analysis for in-hospital cardiac patients. Holter devices collect cardiac information from patient monitors and perform a quantitative analysis not traditionally performed in the monitoring environment. Because MARS VSL directly links into the GE Healthcare CARESCAPE CIC Pro — a central station monitor that assimilates real-time and historical patient data from multiple monitoring sources — a quantitative Holter analysis can now be immediately performed from any connected patient bed, without the need for locating and attaching additional monitoring equipment. Data can then be collected and analyzed anytime, anywhere and for any connected patient bed, then archived to enhance future clinical decision-making.
It leverages the clinical heritage of Marquette clinical monitoring and risk-detection analysis programs. Hospitals can use MARS VSL with existing cardiac technologies to enhance clinical workflow. It enables integration with systems such as the CARESCAPE Monitor B850 bedside monitor that directly links clinical users to electronic medical records (EMRs), diagnostic images, lab results and real-time patient monitoring data, to support efficient clinical decision-making. It also connects to the MUSE Cardiology Information System that facilitates ECG analysis and comparisons to the patient’s prior ECG.
The device is currently available for purchase in the U.S. The sleep apnea detection integration is initially available to U.S. medical centers, and will be rolled out to select European and Asian locations later in the year. Outside the U.S., medical centers can currently purchase MARS Enterprise i, which integrates Holter ECG analysis with vital signs from clinical monitoring, helping to streamline cardiac diagnostics.
For more information: www.gehealthcare.com.
References:
1. Milleran O, et al. European Heart Journal, 2004 May; 25:728-734.
2. V. Kapur, et al. “The Medical Cost of Undiagnosed Sleep Apnea.” SLEEP, Vol. 22, No. 66, 1999.
3. O. Amir, et al. “An Automated Sleep-Analysis System Operated through a Standard Hospital Monitor.” Journal of Clinical Sleep Medicine, Vol. 6, No. 1, 2010.


 March 31, 2025
March 31, 2025