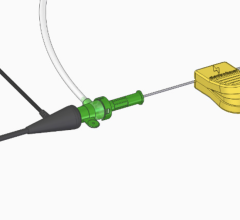
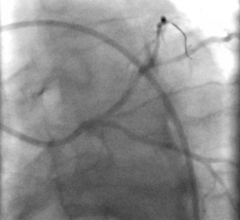
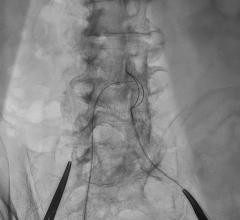
The Merit Laureate hydrophilic guide wire is designed for drainage catheter access, dialysis catheter placement, as well as difficult vascular access procedures.
The guide wire is fabricated from a core nitinol wire, covered with a urethane jacket and coated with a proprietary process that produces a slippery surface. The features of the wire, including torquability and pushability, allow passage through tortuous anatomy.
Merit Medical Systems Inc. announced the product's FDA 510(k) clearance in November 2009.
For more information: www.merit.com


 June 17, 2024
June 17, 2024