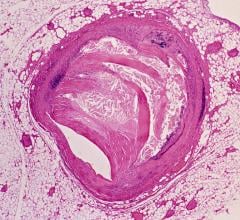
February 8, 2013 — Identive Group Inc. announced new radio frequency identification (RFID) labels for the medical industry that are specially designed to withstand the radiation processes commonly used to sterilize medical devices, equipment and supplies. Unlike standard RFID products that lose their memory content when exposed to radiation, Identive's new radiation-resistant labels enable critical identification, usage and tracking information to reside on medical components and storage containers throughout their useful lives.
RFID technology offers superior convenience and performance to many other identification technologies, such as barcodes, but is just beginning to be adopted for medical applications. One barrier has been the challenge of adapting the technology to operate in harsh and demanding environments, such as sterilization using X-rays or gamma radiation. As an innovator of RFID applications to solve complex problems, Identive has created the new labels to address the stringent manufacturing and operating requirements of the healthcare industry. Identive's new RFID labels are resistant to radiation, offer read/write functionality and have a memory capacity of up to 256 bytes. Compliance with ISO 15693, ISO 18000-3 and EPCglobal(R) standards ensures compatibility with common RFID reader devices worldwide.
For more information: www.identive-group.com

 October 24, 2025
October 24, 2025