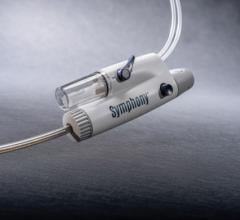
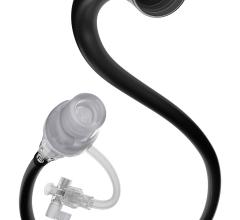
October 21, 2013 — Medtronic Inc. announced the U.S. launch of the Export Advance aspiration catheter, a U.S. Food and Drug Administration (FDA)-cleared catheter that features a pre-loaded stylet that increases deliverability and kink resistance while traversing the vasculature to reach the aspiration site.
The Export Advance aspiration catheter is also constructed with full-wall variable braiding technology, which provides variable degrees of stiffness along the length of the device to enhance flexibility and pushability while navigating the device through the vasculature.
The catheter received CE mark in May and has been introduced in various international markets since then. It is the latest addition to Medtronic’s portfolio of thrombectomy devices, which also includes the Export AP aspiration catheter.
For more information: www.medtronic.com


 March 20, 2026
March 20, 2026