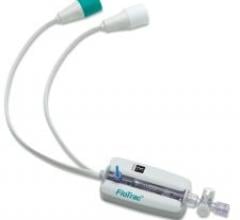
May 14, 2008 - PLC Systems Inc. said it is demonstrating its RenalGuard System May 12-16 at EuroPCR, the annual meeting of the European Association of Percutaneous Cardiovascular Interventions (EAPCI) in Barcelona, Spain.
Many formerly invasive diagnostic and interventional cardiology procedures have migrated to less invasive or percutaneous options. The subsequent rise in cath lab procedures has increased the need for alternatives to manual compression that promote hemostasis at the access site, allowing for quicker patient ambulation and throughput.
May 14, 2008 - Broadlane clients, consisting of approximately 1,100 hospitals, selected ScImage for a three-year ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
Atrial fibrillation (AF) affects more than 3.5 million Americans and is a major source of strokes and a precursor to ...
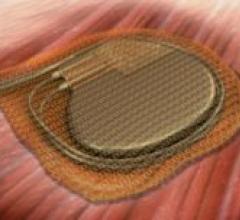
May 13, 2008 - Boston Scientific reported that the FDA cleared its COGNIS cardiac resynchronization therapy ...
The FDA cleared GE Healthcare’s new LightSpeed CT750 HD, said to be the world’s first high definition CT scanner that ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
The FDA granted 510(k) clearance for the AIGISRX CRMD Anti-Bacterial Envelope by Tyrxpharma, a cardiac rhythm medical ...
May 13, 2008 - Philips today announced it has reached an agreement to acquire Brazilian-based Dixtal Biomédica e Tecnologia (Dixtal), a Brazilian manufacturer of in-hospital patient monitoring systems, in a move to bolster Philips’ healthcare presence in high-growth emerging markets .
Abbott Vascular’s HI-TORQUE WHISPER guidewire is designed to offer a higher level of support to facilitate device ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
The Edwards Lifesciences unveiled a new software upgrade that allows greater flexibility to trend and analyze the ...
May 13, 2008 - Edwards Lifesciences Corp. launched a next-generation transfemoral delivery system for the Edwards SAPIEN ...
The FDA cleared for marketing a new automated version of diaDexus’ proprietary PLAC Test, an automated immunoassay designed to run on common existing laboratory equipment, including clinical chemistry analyzers from Hitachi, Roche/Hitachi and Olympus, expanding the number of clinical labs and physician offices that are able to offer the PLAC Test.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
TomTec Imaging Systems recently announced that its new CardioArena, the network and imaging solution for cardiology ...
May 13, 2008 – The FDA cleared GE Healthcare’s new LightSpeed CT750 HD, said to be the world’s first high-definition CT ...
Northeast Monitoring received clearance from the FDA to sell its DR200 Series devices with its automatic atrial ...

 May 13, 2008
May 13, 2008