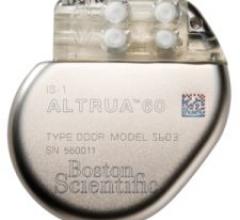
Boston Scientific's ALTRUA family of pacemakers is the first Boston Scientific-branded pacemaker to treat bradycardia ...
May 13, 2008 - Medtronic received FDA premarket approval (PMA) for the first wave of cardiac rhythm disease management ...
Medtronic’s Endeavor Zotarolimus-Eluting Coronary Stent System is engineered for the treatment of coronary artery disease. Endeavor uses the Driver bare metal cobalt alloy stent platform and the drug zotarolimus along with the proprietary, biocompatible drug delivery polymer.
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
Edwards Lifesciences Corp. has rolled out a next-generation transfemoral delivery system for the Edwards SAPIEN ...
GE Healthcare launched its new mobile Vivid S5 cardiovascular ultrasound system, created for multiple care areas ...
May 13, 2008 – The FDA approved changes to the U.S. product label for DEFINITY Vial For (Perflutren Lipid Microsphere) ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
Siemens received FDA 510(k) clearance for Artis zeego, a robotic-assisted positioning capability for interventions in ...
May 13, 2008 - Edwards Lifesciences unveiled a new software upgrade that allows greater flexibility to trend and analyze ...
Textronics Inc. received FDA clearance to market its textile-based ECG Electrode for use in general electrocardiograph monitoring and recording procedures, offering patients an alternative to adhesive electrodes and metal wristbands.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
The FDA cleared CryoLife’s CryoValve SG pulmonary human heart valve processed with the company’s proprietary SynerGraft ...
May 9, 2008 – Philips started shipping the upgrade Xcelera R2.2, a new version of the Xcelera multimodality cardiology ...
May 9, 2008 – The Centers for Medicare and Medicaid Services (CMS) issued its final National Coverage Decision (NCD) to ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
May 9, 2008 - The FDA cleared Boston Scientific's ALTRUA family of pacemakers, a day after the European approval of ...
The Strada Carotid Guiding Sheath is designed specifically to provide easier and faster access to challenging carotid anatomy.
May 9, 2008 – The FDA approved Covidien’s contrast delivery system with radio-frequency identification (RFID) technology, integrating the technology to create a system that is designed to enhance patient safety by reducing the risk of medical errors in radiology departments.

 May 12, 2008
May 12, 2008