In January the FDA granted Ethicon Inc. an expanded indication for EVICEL Fibrin Sealant (Human), which is one of the ...
February 21, 2008 - Textronics Inc. received FDA clearance to market its textile-based ECG Electrode for use in general ...

Feds Helping Boost Technology in Rural America Via Telemedicine
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
Picis will display its CareSuite Extelligence OR, a Web-based business intelligence (BI) solution designed specifically ...
Pinyons Medical Technology Inc. received FDA clearance to market its PowrSyringe Aspirator, a disposable hand-held large ...
Arista AH absorbable hemostasis powder is indicated for most surgeries including cardiac, thoracic, orthopedic, spinal ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...

There are several patient warming and cooling devices available for temperature management, but the practice of induced ...
RES-Q Perioperative Resource Management assesses and confirms surgeons� procedure-specific preferences, staff and equipment requirements, supply lists and inventory and operating room availability, and then sets surgery schedules that aim to diminish resource conflicts and boost productivity.
AVT, a new advanced visualization solution from Barco, reportedly answers the demand for instant access to 3D anywhere, anytime. With AVT, doctors can read volumetric studies throughout the hospital, in a remote office or even at home.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
ARJO offers a full line of sliding surfaces including bariatric Maxi Slide sheets, for bed boosting and lateral ...
February 21, 2008 – McKesson has formed a strategic relationship with Proventys Inc., a personalized medicine service ...
Siemens Medical Solutions received FDA 510(k) clearance for a 1.5T magnetic resonance imaging (MRI) system that is ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...

February 2008 - The traditional hospital cleaning methods of wiping down surfaces and mopping floors are now being ...
February 20, 2008 - Philips Medical Systems features at HIMSS 2008 enhancements to its speech recognition solution ...
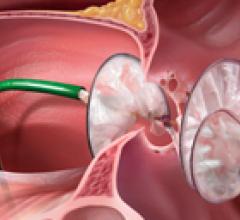
W. L. Gore & Associates (Gore) announced that the FDA granted approval for the use of GORE HELEX Septal Occluder with a modified catheter delivery system indicated for the transcatheter closure of atrial septal defect (ASD), providing a percutaneous ASD closure solution for very young patients.

 February 20, 2008
February 20, 2008