The U.S. Food and Drug Administration (FDA) has approved the Xarelto Starter Pack for deep vein thrombosis (DVT) and/or pulmonary embolism (PE) treatment. The starter pack was designed to help simplify the dosing for the initial 30-day treatment period when patients are at greatest risk of having another DVT or PE.
Biotronik announced that results of its BIOLUX P-I clinical study have been published in the Journal of Endovascular Therapy. BIOLUX P-I was a prospective, international, multi-center, first-in-human, randomized, controlled trial that enrolled 60 patients at five centers in Germany and Austria. The study assessed the safety and efficacy of the Passeo-18 Lux drug-coated balloon (DCB) in treating de novo and restenotic femoropopliteal lesions. The positive results were instrumental in gaining CE mark in December 2013.
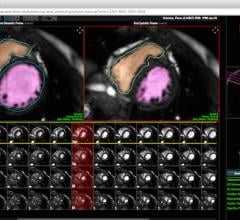
TomTec Imaging Systems GmbH introduced AutoStrain software for automated strain analysis and quantitative visualization of left ventricular function at the 64th Annual Scientific Session of the American College of Cardiology (ACC 2015) in San Diego.
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
The Centers for Medicare & Medicaid Services (CMS) is working closely with stakeholders across the healthcare industry to provide support in transitioning to version 10 of the International Classification of Diseases (ICD), including an online resource. CMS will officially transition from ICD-9 to ICD-10 on Oct. 1, 2015.

Tryton Medical Inc. announced that results from the company’s pivotal clinical trial for the Tryton Side Branch Stent were published in the Journal of the American College of Cardiology. The Tryton Pivotal IDE Trial, which enrolled 704 patients at 67 centers in North America and in 11 countries in the European Union (EU) and Israel, was the largest coronary bifurcation study ever conducted.
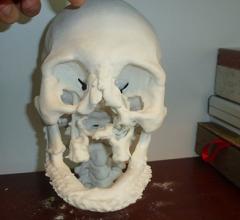
A study being presented at the Society of Interventional Radiology’s annual scientific meeting says 3-D printing could become a powerful tool in customizing interventional radiology treatments to individual patient needs.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
ScImage announced the immediate availability and deployment of updated quantification metrics based on guidelines published jointly in January 2015 by the American Society of Echocardiography (ASE) and the European Association of Cardiovascular Imaging (EACVI).
Heart Imaging Technologies announced the release of Precession, a cardiac magnetic resonance solution which allows viewing, analysis and reporting all within a standard web browser.

Johns Hopkins cardiologists report they have developed a formula that estimates one’s risk of dying over a decade based on a person’s ability to exercise on a treadmill at an increasing speed and incline.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
New and updated American College of Radiology (ACR) Appropriateness Criteria now help healthcare providers choose the most appropriate medical imaging exam or radiation therapy for more than 1,000 clinical indications. These continually updated criteria are a national standard developed by expert panels of physicians from many different medical specialties.
CompView Medical (CVM) introduced an all-in-one equipment manager, visualization and ergonomic boom system — the NuCart.

Frequent use of lead aprons in the interventional lab and radiology departments to protect against radiation exposure is associated with increased musculoskeletal pain, according to a study published in the Journal of the American College of Cardiology.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...

Cardinal Health announced plans to acquire Johnson & Johnson’s Cordis business, a leading global manufacturer of cardiology and endovascular devices, for $1.94 billion in cash.
In a new report, SmarTech Markets Publishing forecasts shipments of 3-D printers in the medical industry will reach 2,135 units in 2020, growing to 3,055 units in 2024. The report, "3-D Printing in Medical Markets: An Opportunity Analysis and Ten-Year Forecast," shows where opportunities will be found.
Hitachi Aloka Medical America Inc. announced the creation of a new dedicated sales force targeting cardiovascular ultrasound opportunities in physician’s offices and hospitals.


 March 04, 2015
March 04, 2015