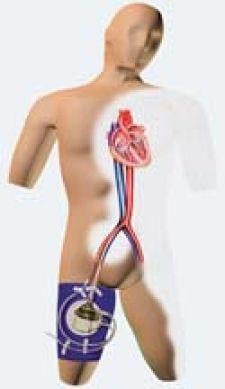
CardiacAssist Inc.'s TandemHeart PTVA in action.
Give a highly ranked research and teaching facility access to cutting-edge technology, and the results could be death defying.
At least that’s true for the Texas Heart Institute (THI) of St. Luke’s Episcopal Hospital (Houston, TX) and its research with the latest in percutaneous left ventricular assist devices.
Mortality rates have been cut by as much as 80 percent nationwide with the TandemHeart PTVA (CardiacAssist Inc., Pittsburgh, PA), making its predecessor obsolete, according to THI cardiologist and heart failure specialist Reynolds M. Delgado, M.D.
THI is one of the first institutions to implant the device, which received FDA clearance a little over two years ago, and is using it more frequently than any other institute in the world — 45 cases in 2005 alone.
Dr. Delgado calls it a “dramatic lifesaver.”
“We have put the device in patients who are on death’s door, supported them, reversed their organ failure and got their bodies back in shape,” he said.
By connecting an external pump to the circulatory system via input and output cannulas, the device supplements the heart’s pumping action, improving blood flow until the patient is strong enough to undergo surgery or angioplasty.
It has also been successful in supporting circulation during high-risk coronary interventions, according to Dr. Delgado.
“High-risk stenting, for example,” he said. “The TandemHeart allows us to do that on an awake patient, without putting him under anesthesia, and we can support circulation while the left main stent is being done.”
Hospital of 1,000 Heart Implants
THI’s success with the device demonstrates its mission of reducing the toll of cardiovascular disease through programs in research, education and patient care.
“It’s a unique model,” said Dr. Delgado. “As a research and teaching entity, it causes a draw for the best and brightest minds in heart disease care.”
Founded in 1962, THI is one of the first standalone cardiovascular institutes in the nation and has been ranked one of the top 10 institutions by U.S. News and World Report for the past 15 consecutive years.
“It’s the first of its kind in this part of the country,” Dr. Delgado said. “So over the years it has really established a strong history of excellence in cardiovascular care.”
The hospital is known for being a pioneer in both artificial and human heart implants.
“We are one of the first centers to implant artificial hearts,” he said. “They are getting smaller and smaller — we now have a pump device that can be implanted in the heart that is as big as a D cell battery that can pump all the blood supply requirements of the body.”
That device is the Jarvik 2000, a valveless, electrically powered miniature axial flow pump that fits directly into the left ventricle and pushes oxygenated blood throughout the body. It is still in experimental stages.
Denton A. Cooley, M.D., THI president and surgeon-in-chief, performed the first successful heart transplant in the U.S. in 1968 and THI chief of cardiovascular transplantation and director of surgical research Bud Frazier, M.D., and his team completed their 1,000th heart transplant this year.
"The devices we've been developing and testing are either ventricular assist devices or artificial hearts,” Dr. Frazier said. “They are all pumps of some type, but they each work differently for a specific function."
Vision of Education
THI’s Heart Information Center provides educational information related to the prevention, diagnosis and treatment of cardiovascular disease.
“One of our strongest visions is education,” Dr. Delgado said. “We have several educational seminars — we are actually in the process of developing one specifically for the TandemHeart.”
Dr. Delgado says the training programs are aimed toward long-term education.
“It’s a wonderful experience for the doctors in training to be able to learn cutting-edge technology,” he said. “Then, wherever they end up practicing, they can take that knowledge with them.”
The institute also has programs ranging from continuing medical to children’s education.
Project Heart is a Web site focused on teaching the basics of the cardiovascular system and cardiovascular health to elementary school children. The program extends the community outreach mission to educators by offering comprehensive curriculum materials from kindergarten through third grade.




 July 02, 2024
July 02, 2024 









