
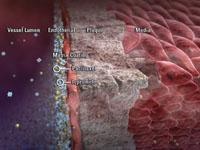
Invatec developed the FreePac balloon coating, which uses a urea matrix to free and separate paclitaxel molecules and facilitates their absorption into the blood vessel wall.
Drug-eluting balloons (DEBs) may offer new options in treating peripheral vessels and restenosis. DEBs already offer an alternative treatment tool in Europe, and the first DEB clinical trial in the United States is expected to begin later this year.
In early 2010, Medrad Interventional/Possis asked the U.S. Food and Drug Administration (FDA) to allow an investigational device exemption (IDE) trial for its Cotavance paclitaxel-eluting balloon. The company is seeking both CE mark and FDA clearance for the device.
If cleared, the trial may begin in late 2010 or early 2011, said William A. Gray, M.D. director of endovascular services, Center for Interventional Vascular Therapy, New York-Presbyterian Hospital/Columbia University Medical Center. He will serve as a co-principal investigator for the trial, which will focus on the use of DEBs in peripheral artery disease (PAD). If started in late 2010 and successful, it might lead to an FDA clearance by 2014.
Invatec, which released four DEBs in Europe last year, is also in initial discussions with the FDA to define the requirements for a U.S. trial of its DEBs, the company said.
“It can really change the way we perform angioplasty,” said Juan F. Granada, M.D., medical director of the Skirball Center for Cardiovascular Research at the Cardiovascular Research Foundation. He has worked with paclitaxel-eluting balloons in preclinical animal studies. “There was a lot of skepticism about the possibility of transferring drugs and maintaining levels into the vessel wall using a single balloon inflation, but I have to tell you there is evidence that suggests it is possible. Ten years ago, no one would buy the concept, but now we are convinced this can be done and I think it has opened a new avenue in interventional cardiology.”
The Need for DEBs
DEBs are expected to play a big role as a new treatment option in the superficial femoral artery (SFA), iliacs and the popliteals, where current interventions are not particularly successful and restenosis is common. “It may be something that really gives a significant treatment option for the popliteals or SFA,” Dr. Gray told Diagnostic & Invasive Cardiology.
“Stents are very versatile, but they have their limitations,” said Dr. Granada. Stents have not proven to be an ideal option in treating long-diffuse disease, small vessels, the infrapopliteals, or in lesions below the knee. “In the end, it’s all about the long-term patency,” Dr. Gray said. He said there is a fair amount of restenosis in these vessels. “We rarely have any issues getting the vessel open, but we have had issues keeping them open.”
The SFA can be up to 36 inches long and is constantly exposed to compression and torque, he explained. The lesions in these vessels also tend to be very long and frequently calcified. Many SFA lesions also are chronic total occlusions (CTOs) that obstruct the vessel from the hip to the knee. “There are not many 2 cm lesions out there. Most are moderate to long lesions,” Dr. Gray said.
Stents and stand-alone balloon angioplasty are used, but restenosis rates are high. Dr. Gray said the longer the lesion, the higher the rate of restenosis. Atherectomy is another option, but long-term patency is not always guaranteed. He said one proposed solution is to combine atherectomy with DEBs for better long-term outcomes.
“I don’t think that in the coronary territory people will go back to angioplasty alone,” Dr. Granada said, even with DEBs. However, he thinks DEBs might have niche applications, such as a stand-alone therapy to treat diffused, long lesions in the legs.
He said that for mainstream coronary applications, the most likely use for DEBs will be as a companion therapy with stents. The DEB could be used to pretreat the vessel wall before the expansion of a bare metal stent. This strategy could potentially offer the benefit of a DES without the polymers that are often blamed for late stent thrombosis. “That is the biggest impact this technology could potentially have,” Dr. Granada said.
PAD is the primary focus for DEBs. “There are different reasons for this, mainly because it is an unmet clinical need,” Dr. Granada said. There is a strong clinical need for better peripheral treatments because of the outcomes from current treatments. DEBs are also expected to be much less expensive than stenting, especially in situations in which numerous stents are needed for longer lesions, he said.
“The data [also] seems to show that it looks like a great application to treat in-stent restenosis,” Dr. Granada said.
The Chemistry Involved
All DEBs in development or already available overseas use the antiproliferative drug paclitaxel, although each company has a different formulation, said Dr. Granada. The drug is loaded onto the balloon with or without a carrier. Dr. Gray said a key advantage of paclitaxel is its lipophilic properties, allowing it to absorb into plaque. Dr. Granada added it not only absorbs easier, but also is retained for a longer period of time compared to other agents. He said limous drugs do not seem to be retained within the plaque as easily, so they may be challenging agents to work with for this particular application.
“Most DEBs are developed to maximize delivery and minimize loss [of the drug],” Dr. Gray said. He said the secret to DEBs is in the carrier mechanism holding the drug on the surface of the balloon. It needs to protect it from dissolving in the blood in transit to the lesion and still offer a fast elution rate once the balloon is expanded.
The Paccocath Matrix
Medrad’s Cotavance uses the Paccocath technology, a balloon coating matrix created by Bayer AG, which owns Medrad. This same technology was licensed to B. Braun for its SeQuent Please coronary DEB released last year in Europe. In February, Medrad and B. Braun announced an agreement to
co-promote the Paccocath technology as Medrad prepares for future marketing of its device.
The Paccocath matrix consists of paclitaxel and the radiologic contrast agent Ultravist 370. The matrix helps evenly distribute and elute the drug when the balloon is inflated. Clinical results to date (including the THUNDER, PEPCAD, ISR and FEMPAC trials) show the Paccocath technology prevents restenosis and late lumen loss over time compared to standard therapies.
The Paclitaxel-Eluting PTCA-Balloon Catheter in Coronary Artery Disease II-In-Stent Restenosis (PEPCAD II-ISR) study compared the SeQuent Please to the Taxus DES in 131 patients over six months. The study showed patients treated with the DEB experienced only 3.7 percent restenosis and 4.8 percent major adverse cardiac events (MACE). This was compared to patients with a DES restenosis rate of 20.8 percent with 22 percent MACE.
The FreePac Matrix
Invatec uses the FreePac coating. The urea matrix frees and separates paclitaxel molecules and facilitates their absorption into the wall of the blood vessel. Invatec President Jack Springer said the carrier agent allows for the balance of hydrophilic and lipophilic properties. “Urea may offer an advantage over the introduction of an additional foreign agent – contrast medium – into the vessel wall,” he said.
Several clinical trials have been initiated by Invatec to assess its DEBs. In January Invatec released the first clinical results from its In.Pact Amphirion trial for complex, critical limb ischemia (CLI) below the knee. Preliminary results indicate a dramatic reduction in restenosis rate by application of the DEB. The standard treatment resulted in 69 percent of CLI patients with long lesions having restenosis after three months. The DEB brought the rate down to 31 percent.
Future Research
Different drugs may be tested for DEBs in the future, most likely limus derivates, said Dr. Granada. In addition, more research will explore new drug carriers and transfer options. He said one area of interest is with local drug delivery carriers, which might allow the drug to remain encapsulated during delivery to the lesion and offer rapid transfer of the drug once the balloon is expanded.
There also are still lingering issues with the drug transfer that need to be addressed. Dr. Granada said DEB drug delivery may be more erratic than drug-eluting stents (DES). The balloon coating also can be sent downstream, which could potentially be an issue.
Competition With Peripheral DES
While DEBs show promise for better outcomes in peripheral vessels, both Drs. Gray and Granada said a new peripheral DES also has the potential to provide better PAD outcomes. They are looking forward to Cook Medical’s Zilver PTX paclitaxel-eluting stent. The stent has demonstrated positive, preliminary clinical results to improve long-term PAD outcomes.
Read the article "Drug-eluting Balloons Enter the U.S. Market."





 June 13, 2024
June 13, 2024 









