
The first pig heart to be transplanted into a human is moved from a perfusion transport system to the patient during the groundbreaking procedure Jan. 7, 2022. All photos from the University of Maryland Medical Center.
January 10, 2022 — The University of Maryland School of Medicine (UMSOM) and the University of Maryland Medical Center (UMMC) announced today the first successful transplant of a genetically modified pig’s heart into a human patient. The heart transplant patient, a 57-year-old Maryland man, is doing well following the groundbreaking cardiovascular surgery on Friday, Jan. 7 to save his life.
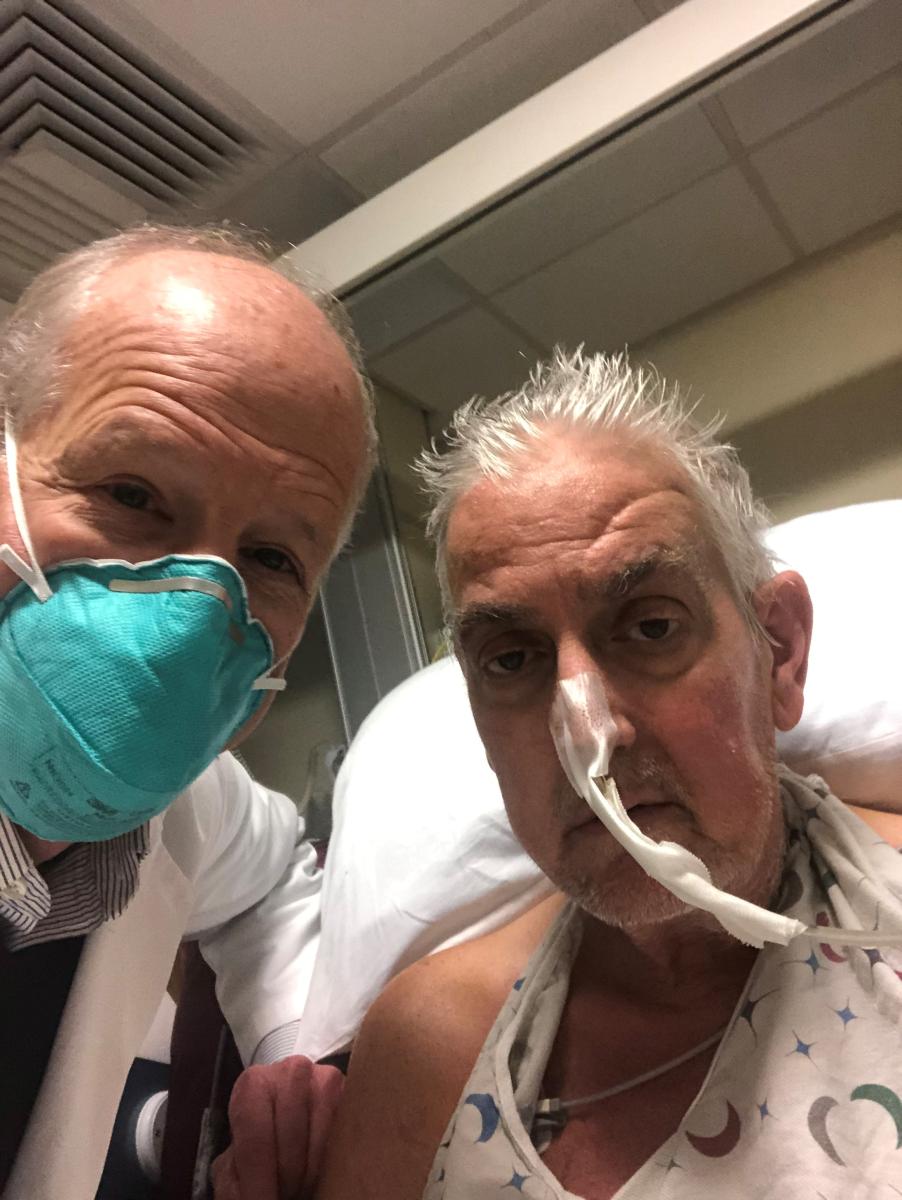
This organ transplant demonstrated for the first time that a genetically-modified animal heart can function like a human heart without immediate rejection by the body. The patient, David Bennett, is being carefully monitored over the next days and weeks to determine whether the transplant provides lifesaving benefits.
He had advanced heart failure and had been deemed ineligible for a conventional heart transplant at UMMC as well as at several other leading transplant centers that reviewed his medical records. He was on ECMO for life support and was not expected to leave the hospital alive.
“It was either die or do this transplant. I want to live. I know it’s a shot in the dark, but it’s my last choice,” Bennett said a day before the surgery was conducted. He had been hospitalized and bedridden for the past few months. “I look forward to getting out of bed after I recover.”
Porcine (pig) heart transplants are not approved by the U.S. Food and Drug Administration (FDA), however, the federal agency authorized the surgery in this case for “compassionate use” as no other options remained for the patient.
The FDA granted an emergency authorization for the surgery on New Year’s Eve through its expanded access (compassionate use) provision. It is used when an experimental medical product, in this case the genetically-modified pig’s heart, is the only option available for a patient faced with a serious or life-threatening medical condition. The authorization to proceed was granted in the hope of saving the patient’s life.
“This was a breakthrough surgery and brings us one step closer to solving the organ shortage crisis. There are simply not enough donor human hearts available to meet the long list of potential recipients,” said Bartley P. Griffith, M.D., who surgically transplanted the pig heart into the patient. Griffith is the Thomas E. and Alice Marie Hales Distinguished Professor in Transplant Surgery at UMSOM. “We are proceeding cautiously, but we are also optimistic that this first-in-the-world surgery will provide an important new option for patients in the future.”
Revivicor, a regenerative medicine company based in Blacksburg, Va., provided the genetically modified pig to the xenotransplantation laboratory at UMSOM. On the morning of the transplant surgery, the surgical team, led by Griffith and Muhammad M. Mohiuddin, M.D., professor of surgery at UMSOM, removed the pig’s heart and placed it in the XVIVO Heart Box perfusion device, which kept the heart preserved until surgery.
The physician-scientists also used a new drug along with conventional anti-rejection drugs, which are designed to suppress the immune system and prevent the body from rejecting the foreign organ. The new drug used is an experimental compound made by Kiniksa Pharmaceuticals.
The pig heart was also genetically modified in order to reduce the chance of rejection, which is an issue with both human heart transplants and previous attempts to use animal heart transplants.
"We have modified 10 genes in this pig heart," explained Mohiuddin. "Four genes were knocked out, three of them responsible for producing antibodies that causes rejection, and then one gene was knocked out to control the growth of the pig and its organs."
He said this should make the hearts more acceptable to the human patient's immune system to prevent rejection.
"This is a game changer because we will now have these organs readily available, if this works," Mohiuddin said. "We have the technique of genetically modifying the hearts, so if we need more tweaking required to modify the genes we will be able to do that and we can customize the heart or the organ for the patient."
The use of porcine hearts for humans has been discussed for years. The anatomy and size of the pig hearts is very similar to human hearts, which is why these animals are used for preclinical testing of new cardiovascular devices. Pig heart valves have also been used for replacing valves in humans for years.
If modified porcine hearts can be shown to be a viable option for transplant into human heart patients this will open the door a new source of widely available donor hearts.
Pig Heart Transplants Into Humans Could Solve Shortage of Human Hearts
This medical breakthrough may help healthcare professionals solve the organ shortage crisis that leaves thousands each year without life-saving heart transplants. About 20% of patients on the heart transplant waiting list die while waiting to receive a transplant or become too sick to be good candidates for the complex transplant procedure.
In 2019 (most recent data available), the United States recorded the highest number of heart transplants, with 3,552 transplantations performed. As of March 11, 2020, 3,661 people were on the waiting list for a heart transplant, and 52 people were on the waiting list for a heart and lung transplant, according to the American Heart Association's 2021 Heart Disease and Stroke Statistical Update.
Animal organ transplants, or xenotransplants, are extremely rare and can have significant risks to patients as well as bioethical concerns, said the American Heart Association (AHA) in a statement about the procedure. But the society added that there is a big need for more donor hearts to match the number of patients on the transplant waiting lists.
The lack of donor hearts or issues some patients have to qualify for the transplant list has led to the use of implantable left ventricular assist devices (LVADs) being used as a bridge-to-transplant, or as permanent destination therapy for some patients. The shortage of hearts also has led to a few vendors developing total mechanical artificial hearts.
According to the AHA, the most common reasons for a heart transplant are that one or both ventricles of the heart are not functioning properly so that severe heart failure is present. A heart transplant gives a patient the opportunity to have a normal heart with normal blood circulation. If the transplant goes well, heart function and blood flow will be closer to normal, and life-sustaining.
Traditionally, a donor heart is matched to the recipient by blood type and body size. Heart transplant recipients must take medications to prevent their immune system from rejecting the new heart. These medicines are called immunosuppressives because they prevent rejection of the new heart by suppressing the body’s normal immune response to a foreign object. However, these drugs may have side effects, which can include an infection or an increased risk of some cancers.
Pig Heart Transplant is Culmination of the University of Maryland Xenotransplantation Program
The University of Maryland School of Medicine has been working toward this type of transplant for several years. Considered one of the world’s foremost experts on transplanting animal organs, Mohiuddin joined the faculty five years ago and established the Cardiac Xenotransplantation Program with Griffith. Mohiuddin serves as the program’s scientific and program director and Griffith as its clinical director.
“This is the culmination of years of highly complicated research to hone this technique in animals with survival times that have reached beyond nine months," explained Mohiuddin. "The FDA used our data and data on the experimental pig to authorize the transplant in an end-stage heart disease patient who had no other treatment options. The successful procedure provided valuable information to help the medical community improve this potentially life-saving method in future patients.”
Xenotransplants were first tried in the 1980s, but were largely abandoned after the famous case of Stephanie Fae Beauclair (known as Baby Fae) at Loma Linda University in California. The infant, born with a fatal heart condition, received a baboon heart transplant and died within a month of the procedure due to the immune system’s rejection of the foreign heart.
Before consenting to receive the transplant, Bennett was fully informed of the procedure’s risks, and that the procedure was experimental with unknown risks and benefits. He had been admitted to the hospital more than six weeks earlier with life-threatening arrhythmia and was connected to an extracorporeal membrane oxygenation (ECMO) machine for to keep him alive. In addition to not qualifying to be on the transplant list, he was also deemed ineligible for an artificial heart due to his arrhythmia.
VIDEO: Details on the First Pig Heart Transplant Surgery in a Human
Read more on this first case: www.medschool.umaryland.edu/news/2022/University-of-Maryland-School-of-Medicine-Faculty-Scientists-and-Clinicians-Perform-Historic-First-Successful-Transplant-of-Porcine-Heart-into-Adult-Human-with-End-Stage-Heart-Disease.html
Muhammad Mohiuddin, M.D., with the team that performed the first pig heart transplant into a human.
Related Heart Transplant News:
Transplanting Pig Hearts Into Humans One Step Closer
FDA OKs Use New Version of Carmat Artificial Heart in U.S. Early Feasibility Study
Carmat Artificial Heart Shows Promising Automatic Regulation of Blood Flow
Heart in a Box Technology Expands Heart Transplant Window
FDA Clears SynCardia 50cc Temporary Total Artificial Heart as Bridge to Transplant
Stephanie’s Heart: The Story of Baby Fae
Hearts from donors who used illicit drugs or overdosed safe for transplant, cuts wait time
National Trends in Heart Donor Utilization Rates: Are We Efficiently Transplanting More Hearts?
Heart transplants from donors with hepatitis C may be safe and could help decrease organ shortage


 May 13, 2026
May 13, 2026 









