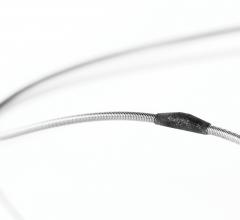
Nov. 5, 2014 — Food and Drug Administration gave 510(k) clearance for the HawkOne directional atherectomy system. The ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
November 4, 2014 — Shockwave Medical announced that the company will present clinical results from DISRUPT PAD, a single ...
November 4, 2014 — The U.S. Food and Drug Administration (FDA) has cleared the Abbott family of Xience everolimus ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
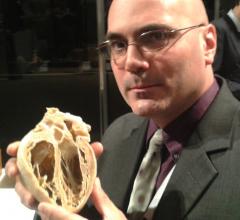
November 3, 2014 – Once a common procedure, left ventriculography’s role in assessing heart disease has evolved ...
By Dave Fornell, editor of DAIC Magazine
The key take away messages from the 26th annual Transcatheter Cardiovascular ...
October 31, 2014 — Metro Health (Michigan) cardiovascular specialist Jihad Mustapha, M.D., is one of the first ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
October 30, 2014 — Cardiovascular Systems Inc. announced that the first seven patients in Japan have been enrolled in ...
October 29, 2014 — Restenosis, the recurrence of narrowing of the arteries after stenting, is a common risk of this ...
October 27, 2014 — Miracor Medical Systems announced today the successful launch of its next-generation Pressure ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
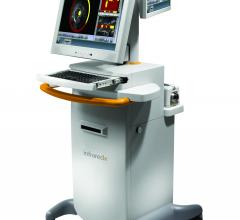
October 27, 2014 – Infraredx Inc. today announced that it has entered into an agreement with HealthTrust, a group ...
October 27, 2014 — Merit Medical Systems Inc. recently unveiled its new ThinkRadial website and educational initiative ...
Oct. 27, 2014 — The U.S Food and Drug Administration (FDA) cleared the EverFlex Self-Expanding Peripheral Stent System ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
October 24, 2014 — The quality and resolution of X-ray images depends on the characteristics of the focal point, the ...
October 23, 2014 — The use of stents has improved management and outcomes of coronary artery disease, and clinical ...
October 21, 2014 — Siemens Healthcare has announced that the U.S. Food and Drug Administration (FDA) has cleared the new ...

 November 05, 2014
November 05, 2014