September 11, 2007 – Improving how healthcare providers follow proven protocols could reduce the risks of subsequent ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
September 11, 2007 - Stereotaxis Inc. said more than 10,000 procedures have been performed using the Stereotaxis ...

When it comes to diagnosing disease, image is everything. It is difficult to treat what you can’t see. That’s ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
Just when drug-eluting stents (DES) were going to revolutionize interventional cardiology, new reports that DES ...
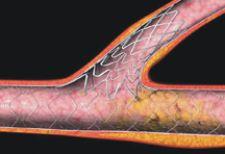
iffering approaches for the treatment of coronary bifurcation and ostial lesions (CBOL) continue to feed the ...
September 7, 2007 - Gary Ansel, M.D., Riverside Methodist Hospital, successfully implanted the GORE VIABAHN ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
September 10, 2007 - Four analyses of three-year follow-up data that were presented at the ESC Congress 2007 showed that ...
Possis Medical Inc. will feature the newest advancements in its “Complete Solution for Thrombus,” including the ...
Cordis Endovascular announced, at the 61st Annual Meeting of the Society for Vascular Surgery, the worldwide launch of ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
September 6, 2007 - Cleveland Clinic will host its fifth annual Medical Innovation Summit, an international ...
September 6, 2007 – Boston Scientific Corp. announced at the ESC 2007 conference in Vienna, Austria, that results from ...
September 5, 2007 – According to Boston Scientific Corp., results from the TAXUS WOMAN study, a gender specific analysis ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
September 5, 2007 - The National Institutes of Health has awarded its $5 million Bioengineering Research Partnership ...
September 5, 2007 - Clinical investigators at the European Society of Cardiology Congress 2007 (ESC Congress 2007) ...
September 4, 2007 - Analysis of clinical trials showed the Cypher Sirolimus-eluting coronary stent was associated with ...

 September 10, 2007
September 10, 2007




