February 12, 2010 - After the U.S. FDA announced it is launching a program designed to reduce unnecessary medical ...
PET Imaging
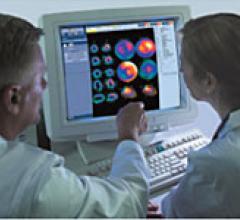
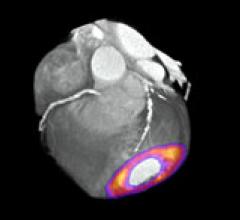
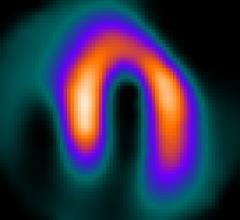
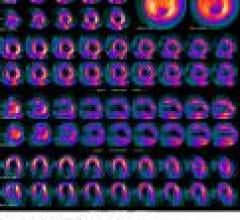
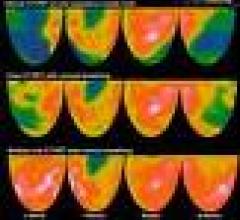
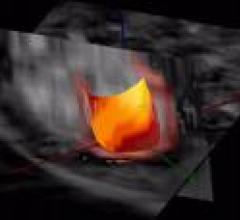
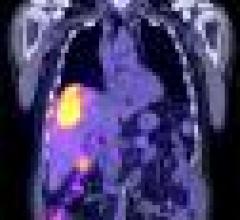
Positron emission tomography (PET) is a nuclear imaging technology (also referred to as molecular imaging) that enables visualization of metabolic processes in the body. The basics of PET imaging is that the technique detects pairs of gamma rays emitted indirectly by a positron-emitting radionuclide (also called radiopharmaceuticals, radionuclides or radiotracer). The tracer is injected into a vein on a biologically active molecule, usually a sugar that is used for cellular energy. PET systems have sensitive detector panels to capture gamma ray emissions from inside the body and use software to plot to triangulate the source of the emissions, creating 3-D computed tomography images of the tracer concentrations within the body.
February 2, 2010 - Two protocols for reduced-dose cardiac imaging, without compromising image quality, may help ...
January 26, 2010 – The U.S. Department of Energy’s National Nuclear Security Administration (NNSA) selected GE ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
January 25, 2010 - Heart failure is a serious medical condition, in which the heart muscle progressively loses its ...
December 29, 2009 - Imaging is at the heart of diagnostic procedures and is the focus of the "ESC Textbook of ...
December 28, 2009 - The Medical College of Wisconsin will license a novel molecular imaging technology designed ...
November 18, 2009 – The Centers for Medicaid and Medicare Services (CMS) recently announced it will increase the ...

Our profession faces one of the most significant crises in its history,” said Michael Graham, M.D., president of ...
November 12, 2009 - Positron Corp. predicts an industry wide demand for cardiac PET imaging, as a result of the ...
October 6, 2009 – Siemens Healthcare announced the availability of syngo Dynamic PET with myocardial blood flow, a ...
September 30, 2009 – Positron Corp. predicts a significant rise in demand for cardiac PET in 2010 with a proposed ...
September 29, 2009 - Positron Corp., a molecular imaging solutions company focused on nuclear cardiology, will ...
September 21, 2009 – IBA (Ion Beam Applications S.A.), a provider of PET radiopharmaceuticals, and Aposense Ltd ...
September 17, 2009 - Lantheus Medical Imaging Inc. today announced the initiation of a Phase 1 clinical study to ...
September 15, 2009 - Positron Corp. announced that the Ministry of Health and Long Term Care of Ontario, Canada is ...

 February 12, 2010
February 12, 2010