PO Box 4195
Bloomington , IN 47402-4195
USA
Product Categories:
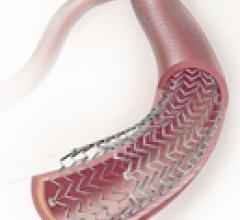
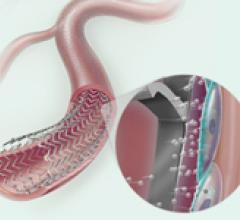
Embolization devices, Stent Grafts, Stents, Stents Bare Metal, Stents Drug Eluting, Stents Peripheral
Embolization devices, Stent Grafts, Stents, Stents Bare Metal, Stents Drug Eluting, Stents Peripheral

 October 21, 2009
October 21, 2009