November 22, 2021 — Atlantic Health System’s Morristown Medical Center announced that it has enrolled the first patient in the world in the PROGRESS clinical trial (“A Prospective, Randomized, Controlled Trial to Assess the Management of Moderate Aortic Stenosis by Clinical Surveillance or Transcatheter Aortic Valve Replacement”), which has been given the approval of the U.S. Food and Drug Administration.
Philippe Genereux, M.D., an interventional cardiologist, and a director of the Structural Heart Program at the Gagnon Cardiovascular Institute at Morristown Medical Center serves as the trial’s principal (lead) investigator.
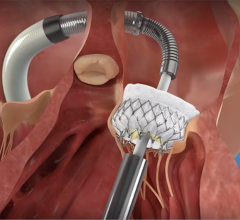
Traditionally, patients with aortic stenosis (AS)—a narrowing of the aortic valve in the heart that keeps it from opening fully, are regularly followed and monitored by their cardiologist, with current cardiology society guidelines recommending valve replacement only when they reach a critical or severe degree of stenosis. However, some patients with moderate AS may develop irreversible heart damage or even die before AS becomes severe. The PROGRESS trial will evaluate whether there is benefit from replacing the aortic valve via a minimally invasive, non-surgical, catheter-based procedure (called a transcatheter aortic valve replacement, or TAVR) before patients progress to a severe degree of AS, as compared to the standard of care of clinical surveillance.
“The PROGRESS trial is extremely important trial for the more than 2.5 million people who suffer from aortic stenosis. It may change the current paradigm of waiting until patients reach a severe or critical degree of stenosis before replacing their valves,” said Dr. Genereux. “The progression of aortic stenosis is unpredictable, and there may be a price to pay for waiting to treat – the goal of early intervention with valve replacement is to preserve the heart’s function, prevent further heart deterioration, and in some cases, prevent death.”
“We are excited to randomize the first patient to the PROGRESS trial,” said Linda D. Gillam, M.D., MPH, the Dorothy and Lloyd Huck chair of Cardiovascular Medicine at Morristown Medical Center/Atlantic Health System. “This illustrates one of the many benefits of research at Morristown Medical Center and Atlantic Health System, giving our patients access to new approaches to their care.”
About the PROGRESS Trial
The PROGRESS trial (A Prospective, Randomized, Controlled Trial to Assess the Management of Moderate Aortic Stenosis by Clinical Surveillance or Transcatheter Aortic Valve Replacement) will enroll patients aged 65 and older who have been diagnosed with moderate aortic stenosis with symptoms or already established signs of cardiac damage. Participants will be randomized to receive a transcatheter aortic valve replacement (TAVR) with the Edwards SAPIEN 3/Ultra heart valve, or standard of care clinical surveillance. Up to 750 patients will be enrolled worldwide. Patients in this study will be followed carefully up to 10 years by a team of heart valve experts.
The PROGRESS trial was approved by the FDA and is sponsored by Edwards Lifesciences.
About Aortic Stenosis (AS) and Transcatheter Aortic Valve Replacement (TAVR)
Aortic stenosis (AS), one of the most common and serious valve diseases, is a narrowing of the aortic valve opening, which restricts blood flow from the left ventricle of the heart to the aorta. Symptoms of AS include shortness of breath, chest pain such as pressure or tightness of the chest, fainting, feelings of a heavy, pounding heart, decreased physical activity or an unusual sound heard between heartbeats known as a heart murmur. TAVR is a minimally invasive procedure to replace an aortic valve that has narrowed and fails to properly open.
About Clinical Trials at the Gagnon Cardiovascular Institute at Morristown Medical Center
Through its Gagnon Cardiovascular Institute, Atlantic Health System’s Morristown Medical Center was the first hospital in New Jersey to offer TAVR for aortic stenosis and treats more valve patients than any other single hospital in New Jersey and the New York area. Morristown Medical Center offers specialized treatment for all types of heart valve disease and structural heart problems. The Valve Center of Morristown recently received the American College of Cardiology (ACC) Certification of Center of Excellence for its exceptional care related to TAVR patients.
About Heart Care at Atlantic Health System
Morristown Medical Center, the hub of heart care at Atlantic Health System, is one of the top 45 programs in the country for Cardiology & Heart Surgery (U.S. News & World Report) and one of America’s 100 Best Hospitals for Cardiac Care (Healthgrades).
Its expert team presents research across the world and is comprised of interventional cardiologists, electrophysiologists, heart failure experts, advanced imagers and cardiac surgeons.
The program also offers specialty care in hypertrophic cardiomyopathy, sports cardiology, cardio-oncology, pediatric cardiology, and heart disease in women. The team works closely with community cardiologists who provide a medical home for patients with heart disease.
Atlantic Health System Heart Care is focused on offering minimally invasive and non-invasive procedures that help patients return to their lives faster. The state’s largest valve replacement/repair program offers valve therapy options for the aortic, mitral, and tricuspid valves, and is helping advance the radial (wrist) approach for coronary procedures with a new lounge for post-procedure recovery.
Morristown Medical Center is home to the largest cardiac surgery and invasive cardiology programs in New Jersey and offers the most up-to-date cardiac imaging technologies available.


 July 08, 2024
July 08, 2024