January 9, 2018 – Avinger Inc. announced that Arne Schwindt, M.D., a vascular surgeon at St. Franziskus Hospital in Münster, Germany, has successfully treated the first seven patients with the next-generation Pantheris Lumivascular atherectomy system. Schwindt, an experienced operator of Lumivascular technology, used the next-generation Pantheris to treat seven separate patients with a variety of lesion types, including in-stent restenosis (ISR), a calcified lesion and a chronic total occlusion, in arteries above and below the knee.
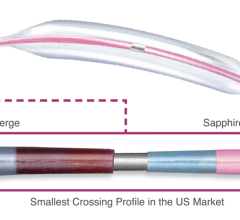
The next-generation Pantheris design includes a simplified single-balloon system for both apposition of the device and occlusion of blood flow, a stiffer shaft for increased pushability, a more robust nose cone with the option for more tissue storage capacity and an enhanced cutter design. This device is available in limited supply for commercial sale in the European Union; it is not available commercially in the United States at this time.
In each of the seven cases, Pantheris performed as intended, and Schwindt made note of the design features that enabled significant performance improvements as compared to previous versions of the device, including better directional control of the cutter blade for more precise atherectomy. Schwindt also provided feedback about the increased maneuverability of the system, greater tissue capture versus the similarly sized previous version of Pantheris, increased efficiency and ease of cleaning the device between insertions, and high-quality imaging in a variety of vessel diameters.
In commenting on his initial cases, which included a patient with a total occlusion of a stent and multiple below-the-knee lesions, Schwindt noted, “Next-generation Pantheris with real-time image guidance enabled me to see the stent struts and precisely target and remove substantial amounts of tissue that had grown into the stent. In two patients the disease extended below the knee. Image-guided atherectomy with the next-generation Pantheris made it possible to precisely target and remove atherosclerotic plaque in this region while conserving the deep wall components and thus reducing the risk of perforations in these small vessels. The angiographic outcome was excellent.”
Atherectomy is a minimally invasive treatment for peripheral artery disease (PAD) in which a catheter-based device is used to remove plaque from a blood vessel. Lumivascular technology allows physicians, for the first time ever, to see from inside the artery during an atherectomy procedure by using an imaging modality called optical coherence tomography (OCT) that is displayed on Avinger’s proprietary lightbox console. Physicians performing atherectomy with other devices must rely solely on X-ray as well as tactile feedback to guide their interventions while treating complicated arterial disease. With the Lumivascular approach, physicians can more accurately navigate their devices and treat PAD lesions, thanks to the real-time OCT images generated from inside the artery, without exposing healthcare workers and patients to the negative effects of ionizing radiation.
For more information: www.avinger.com


 November 08, 2024
November 08, 2024