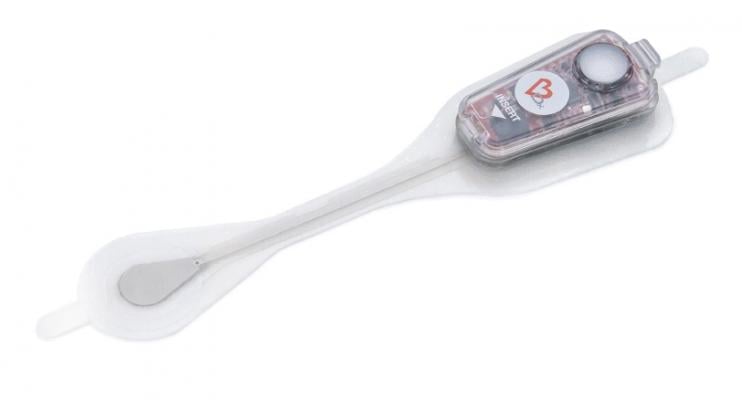
August 15, 2019 — Bardy Diagnostics Inc. announced that HealthTech Arkansas, a healthcare accelerator and investment fund that connects early-stage healthcare companies with disruptive technologies to Arkansas healthcare providers, has selected BardyDx to participate in the organization's 2019 accelerator program. BardyDx was chosen for its advancements in cardiac monitoring by delivering diagnostic accuracy with the Carnation Ambulatory Monitor (CAM), a P-wave centric ambulatory cardiac patch monitor and arrhythmia detection device.
HealthTech Arkansas is a new and expanded iteration of two previous accelerator programs in the state, Health InnovatAR and HubX-LifeSciences. This year, with more provider partners and increased investment for participating companies, HealthTech Arkansas is focused on bringing the most innovative healthcare technologies to Arkansas. The 2019 cohort selection was announced at the Arkansas Heart Hospital with a welcome from CEO Bruce Murphy, M.D., Ph.D., followed by a message from Arkansas Gov. Asa Hutchinson and presentations from each of the six chosen companies. Both Murphy and Hutchinson focused on the importance of working together to bring innovative technologies to Arkansas.
This year's six companies were selected from hundreds of applicants across 18 different countries. The cohort will participate in a program that provides them the opportunity to partner with and build solutions specifically identified by Arkansas Heart Hospital, Arkansas Children's, Arkansas Urology, Baptist Health, CHI St. Vincent, Conway Regional, Mercy, the University of Arkansas for Medical Sciences (UAMS) and Washington Regional Medical Center as areas of opportunity within their organizations. Each of the cohort companies will receive seed investment as well as complete and total access to clinicians and administrators at each partnering site.
"We're delighted to have the participation of BardyDx as one of six companies in our cohort this year," said Jeff Stinson, Director of HealthTech Arkansas. "Collectively, they're among the most accomplished early-stage healthcare companies in the country. BardyDx was specifically chosen because the leadership teams in our partner hospitals believe the company can significantly increase the quality of care through its innovative devices. We can't wait to begin our work with BardyDx."
For more information: www.bardydx.com


 March 04, 2026
March 04, 2026 









