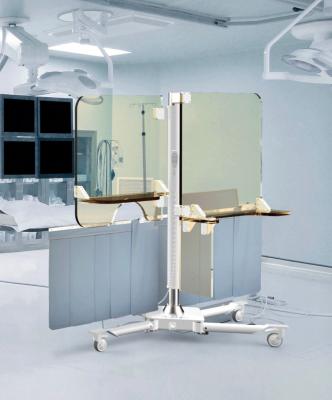
The Rampart iC radiation protection shield for interventional cardiologists in the cath lab.
July 21, 2020 — Interventional cardiology is ranked as the occupation with the highest level of radiation exposure in the world, with providers facing levels that are typically three times higher than those faced by workers in nuclear power plants. As a result, physicians and staff are required to wear lead aprons during surgical procedures, and while this equipment provides some protection, it has a number of drawbacks.
"We wear lead aprons for radiation protection, but they only protect the areas of the body that are covered - namely the torso," said Robert Foster, M.D., FACC, RPVI, an interventional cardiologist at the Birmingham Heart Clinic. "Our brains, our eyes, our arms and any uncovered parts of the body are not protected. I do a lot of peripheral work, and many of our cases can take several hours. As a result, our radiation exposure continues to climb."
In addition to potential health hazards that can result from cath lab angiograpy imaging system radiation exposure, providers are also prone to spinal issues because of the weight of the lead aprons. Statistics from the Society of Cardiac Angiography and Interventions (SCAI) show that when providers work in a catheterization lab for more than 20 years, approximately 60 percent begin to exhibit chronic spinal problems, resulting in the need to reduce case volumes or avoid the lab altogether.
"The aprons weigh between 15 and 30 pounds. Everybody in the lab has to wear one," Foster explained. "For the first 15 years of my career, I was in the lab eight to 10 hours a day wearing about 20 to 30 pounds of lead. I ruptured the first disc in my spine when I was 45, and another disc ruptured five years ago in my early 50s. The second rupture caused paralysis in my leg which resulted in a second surgery. I swore I would never wear a lead apron again."
So Foster, a cardiologist at St. Vincent's East, decided to find a solution. In 2014, he met with several local engineers and together they came up with a revolutionary device that protects providers from radiation without the heavy aprons. They named it the Rampart IC M1128 because a rampart is a barrier that protects a community, thus reducing the need to wear body armor, and M1128 comes from the Bible verse, Matthew 11:28 - "Come to me all who are weary and heavy burdened and I will give you rest."
Tom Livingston, president and COO of Rampart IC, is Foster's partner in the design and an engineer himself. "The Rampart IC is sophisticated but simple. The solution was to transfer the weight of the protection to a support structure and the floor," Livingston said. "The difficulty was engineering a device that is configurable to the many procedures these physicians do and allowing multiple access sites to the body."
The Rampart IC is made of clear, lead-infused acrylic panels that block the radiation from reaching the operator. Physicians and technicians are protected from radiation scatter behind the device. It has a mobile base that allows the device to be moved as needed. Because of this protection, interventionalists may be able to perform more procedures safely and with more comfort.
"Our occupational risks are extremely high, but we are patient-focused and would place ourselves at risk in order to perform our skills in the cath lab," Foster said. "The Rampart IC mitigates the orthopedic risk by reducing the need for heavy aprons. If we can reduce the spine issues caused by the weight on our shoulders, this will be a huge improvement to the cath lab environment for physicians and their teams. Also, it will allow us to help more patients and possibly extend our careers.
"I couldn't have created this by myself. It involved some special people who have a lot of skill. I believe this product will change every lab in the world to the point where we will look back 10 years from now and say we can't believe we wore 30 pounds of lead on our bodies."
Suzannah Campbell, president of St. Vincent's East, praises the work of Foster and his colleagues in the development of the device. "The safety of our patients is always a top priority and so is the safety of our providers," she says. "We have received good feedback about the Rampart IC. The ease of use has had a positive impact on our physicians and radiology techs. I am excited for Dr. Foster and our team."
For more information: RampartIC.com

 October 24, 2025
October 24, 2025