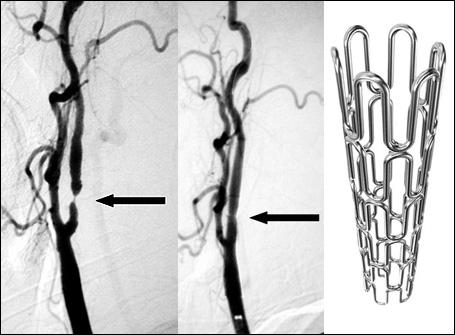
January 19, 2011 – Stents can open up blocked brain arteries after other stroke treatments have failed, according to research presented this week at the 23rd annual International Symposium on Endovascular Therapy (ISET).
In research conducted at the Baptist Cardiac and Vascular Institute in Miami, stents were placed in the blocked brain arteries of 19 acute stroke patients who had not been helped by clot-busting drugs or clot-removal devices. The stents opened up the arteries in 18 of the 19 patients (95 percent), and this resulted in 12 (63 percent) who had minimal or no deficits. Five patients (26 percent) died from major strokes.
If stents hadn’t been used to restore blood flow after these serious strokes, mortality or severe disability would have occurred in approximately 80 to 90 percent of patients, said Italo Linfante, M.D., director of endovascular neurosurgery at the Institute. Currently, using stents to treat acute stroke is an experimental procedure of last resort, used when all other methods have failed.
“FDA-approved treatments to restore blood flow in acute ischemic stroke work 40 to 60 percent of the time. If the artery remains blocked, the patient will suffer death or severe disability,” Linfante said. “Our findings suggest stents can work when clot-busting drugs and clot-removal devices do not, and are a safe and feasible option.”
Stroke is the third-leading cause of death in the United States. Most strokes (85 percent) are caused by a blocked blood vessel, which stops blood flow and oxygen to the brain and causes the brain tissue to die. Treating a stroke quickly can stop further damage, prevent or lessen disability and avert death. Approved treatments include breaking up the clot with drugs, removing it with a corkscrew-like device and vacuuming it out.
Stents have long been used preventatively to open up clogged heart and neck arteries to thwart stroke and heart attack. Scientists have recently discovered that stents may also be used as a treatment device, meaning doctors place them in blocked arteries to treat heart attacks and strokes.
The International Symposium on Endovascular Therapy (ISET) is attended by physicians, scientists, allied professionals and industry professionals from around the world. The meeting pioneered the use of live cases to promote the multidisciplinary treatment of cardiovascular disease. ISET is presented by Baptist Cardiac and Vascular Institute. ISET 2011 is taking place Jan. 16-20 in Miami Beach, Fla.
For more information: www.ISET.org


 May 06, 2026
May 06, 2026 









