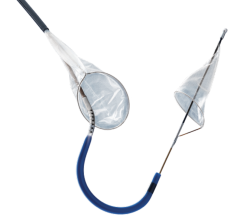
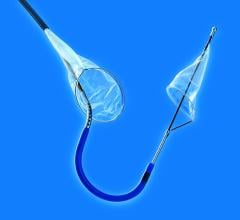
January 19, 2009 - W.L. Gore and Associates today said Peter A. Soukas, M.D., an interventional cardiologist from Caritas St. Elizabeth’s Medical Center in Boston, MA, enrolled the first patient in the Gore EMBOLDEN Clinical Study Jan. 6.
The first patient was recently symptomatic and greater than 90 years old. The greater than 95 percent stenosis in the internal carotid artery was successfully stented under protection by the Gore Embolic Filter. The patient was discharged from the hospital and is doing well.
“In spite of the near total occlusion, the Gore Embolic Filter crossed easily and provided a stable platform for the remainder of the procedure,” Dr. Soukas said. “Moreover, the filter captured an impressive amount of embolic debris. One particularly large particle of plaque that was retrieved by the device would likely have put the patient at risk for a symptomatic event.”
“We are pleased to get this important study underway,” commented William A. Gray, M.D., Columbia University Medical Center, national principal investigator for the Gore EMBOLDEN Clinical Study. “Key device attributes, including low crossing profile, smooth tip transitions and reliable vessel wall apposition, were in evidence and proved beneficial in this challenging case.”
The Gore EMBOLDEN Clinical Study is a prospective, multi-center, nonrandomized, where the Gore Embolic Filter is used for neuroprotection during carotid artery stenting (CAS) in patients at high risk for carotid endarterectomy. The GORE Embolic Filter incorporates a unique diamond frame. The diamond frame is designed to optimize vessel wall apposition and filter efficiency, even in tortuous vessels with small landing zones and tight curves.
For more information:www.goremedical.com.


 April 25, 2023
April 25, 2023