August 6, 2019 — Cardiovascular Systems Inc. (CSI) has acquired the Wirion Embolic Protection System and related assets from Gardia Medical Ltd., a wholly owned Israeli subsidiary of Allium Medical Solutions Ltd.
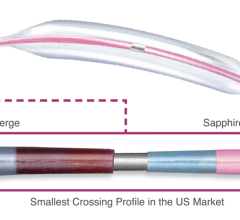
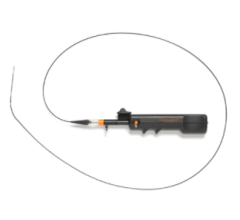
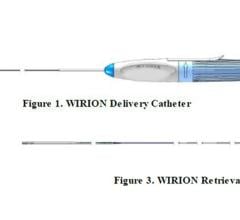
The device, which received European CE Mark in June 2015 and U.S. Food and Drug Administration (FDA) clearance in March 2018, is a distal embolic protection filter used to capture debris that can be associated with all types of peripheral vascular intervention (PVI) procedures. Physicians typically use embolic protection devices in vessels located above the knee with long lesions, high plaque burden and poor run off.
Ehrin Armstrong, M.D., professor of medicine at the University of Colorado, said, “The use of an embolic protection device during peripheral interventions where the risk of embolization is higher provides assurance and protection from procedural complications.”
The Wirion System is easier to use and more versatile than other available embolic protection systems, according to the company, because it can be used with any .014-inch guidewire and for all types of peripheral interventions. In addition, the Wirion System is the only embolic protection device indicated for use with any atherectomy system. The WISE LE study also demonstrated a major adverse event (MAE) rate of 1.9 percent, which is lower than any other previously reported rates with other embolic filters. No clinically significant distal embolization was observed when the Wirion System was used.
WISE LE (Wirion EPS in Lower Extremities Arteries) was a multicenter study, performed in the United States and Germany and included all commercially available atherectomy systems. The primary endpoint was freedom from MAEs occurring within 30 days post-procedure and was compared with an objective performance goal derived from historical atherectomy trials. MAE was defined as a serious adverse event that resulted in death, acute myocardial infarction, thrombosis, pseudoaneurysm, dissection (grade C or greater) or clinical perforation at the filter location, clinically relevant distal embolism, unplanned amputation or clinically driven target vessel revascularization. The study also included a histopathological analysis of debris captured by the filter during the procedures.
CSI plans to commercialize the Wirion System in the U.S. following the transfer of manufacturing from Gardia Medical. CSI expects the manufacturing transfer to be completed after a 12- to 15-month transition period. Gardia will retain the rights to the Wirion System for angioplasty and stenting procedures in the carotid arteries.
For more information: www.csi360.com, www.allium-medical.com
Related Embolic Protection Device Content


 November 08, 2024
November 08, 2024