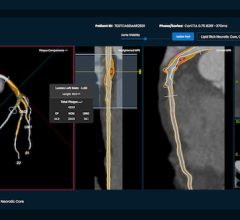
June 4, 2024 — HeartFlow, a leader in cardiovascular healthcare technology, is pleased to announce a key Medicare policy development, which should allow for future expanded patient access to their Plaque Analysis product. Five Medicare Administrative Contractors (MACs), CGS, NGS, Noridian, Palmetto GBA, and WPS, released draft Local Coverage Determinations (LCD) for Artificial Intelligence Enabled CT Based Quantitative Coronary Topography (AI-QCT)/Coronary Plaque Analysis (AI-CPA). These draft LCDs recognize the importance of quantifying coronary artery plaque, which underscores the pivotal role HeartFlow technology plays in diagnosing and managing treatment for patients in need of cardiovascular care.
HeartFlow’s accurate and actionable Plaque Analysis aligns with the Centers for Medicare & Medicaid Services (CMS) commitment to supporting technologies that enhance diagnostic accuracy and patient care:
- HeartFlow Plaque Analysis is the only FDA cleared plaque analysis with a reported 95% agreement prospectively compared to the gold standard, IVUS.¹
- Two out of three patients had their medical management changed and more precisely tailored with HeartFlow Plaque Analysis.²
Plaque Analysis is a major function of the HeartFlow portfolio, which offers the only comprehensive, AI-driven, precision coronary care solution based on the guideline-directed cardiac CT pathway, backed by robust clinical evidence and guidelines. HeartFlow FFRCT has an established reimbursement pathway which is covered for 99% of people in the US with health insurance. Leveraging our expertise and proven success creating new AI reimbursement categories as done with HeartFlow FFRCT, similar coverage and adoption for Plaque Analysis is anticipated, ensuring it becomes an integral part of coronary care.
“We are excited about this milestone, as it highlights the critical importance of accurately quantifying plaque in cardiovascular disease management,” said John Farquhar, chief executive officer at HeartFlow. “This recognition by the MACs in the draft policies is a testament to the clinical value of Plaque Analysis and its potential to revolutionize how cardiovascular disease is diagnosed and treated, for all.”
The release of these draft LCDs mark the first step in integrating accurate plaque measurement into standard clinical practice. Next steps toward coverage include a period for input from clinicians and other stakeholders before policy is finalized and released.
“We are confident that Plaque Analysis will not only meet but exceed the rigorous standards of Medicare,” added Cara Santillo, Senior Vice President of Market Access and Reimbursement at HeartFlow. “This nod of support from the MACs underscores the importance of access and equity in cardiovascular care by ensuring that all patients, regardless of background, receive the critical insights needed for treatment for cardiovascular disease.”
For more information: www.heartflow.com


 April 15, 2026
April 15, 2026