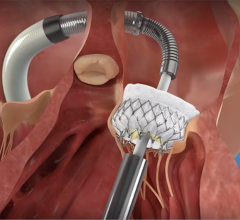
Mitralign's Percutaneous Tricuspid Valve Annuloplasty System uses catheter delivered pledgeted sutures that can be cinched together to change the valve annulus geometry to help eliminate valvular regurgitation.
August 31, 2015 — Mitralign Inc. said the U.S. Food and Drug Administration (FDA) granted an investigational device exemption (IDE) so the company can conduct an early feasibility study to examine its Percutaneous Tricuspid Valve Annuloplasty System (PTVAS). The SCOUT Study will take place in select centers in the United States.
“Percutaneous repair of the tricuspid valve represents one of the largest remaining unmet needs in the structural heart space,” said Rick Geoffrion, chief executive officer of Mitralign. “We mimic a known surgical procedure but do not require open heart surgery, providing a potential treatment option for a large number of patients that are too sick for surgical intervention. This approval from the FDA demonstrates their continued commitment to encouraging medical device innovation to address clinical needs and improve patient care.”
“Even though the first group of patients treated were very sick, compassionate use candidates in Europe, the system has shown an excellent safety profile along with tremendous improvements in significant clinical measures,“ commented Rebecca Hahn, M.D., director of interventional echocardiography, Columbia University Medical Center / New York Medical Center / New York Presbyterian Hospital and principal investigator for the SCOUT study. “This patient population has very limited options, and the possibility of performing a transcatheter repair is very exciting. This innovation has significant potential to be a first line therapy.”
Mitralign is the first company to provide interventional cardiologists with the tools to perform minimally invasive, percutaneous procedures for both functional mitral regurgitation and tricuspid regurgitation with the Mitralign Percutaneous Mitral Repair System expected to receive CE mark approval for treatment of functional mitral regurgitation (FMR) in 2015. Both systems utilize the same innovative platform. The Percutaneous Tricuspid Valve Annuloplasty System and Mitralign Percutaneous Mitral Repair System are under clinical investigation and not available for sale or distribution.
Tricuspid regurgitation (TR) Tricuspid regurgitation occurs when the tricuspid valve fails to open and close properly, causing blood to flow backwards into the right atrium. If left untreated, TR can lead to heart enlargement and heart failure. In the U.S. alone, there are an estimated 1.6 million patients suffering from TR.[1] It is estimated that 50 percent of patients with mitral regurgitation have moderate to severe tricuspid regurgitation.[2] TR is currently undertreated by surgery. In the United States, surgeons treat only 5,500[3] patients per year, most of them in conjunction with left heart surgeries. When treating the valves, surgeons choose repair 90 percent of the time versus replacement (10 percent).[3]
For more information: www.mitralign.com
References:
1. Stuge O., Liddicoat J., et al. JTCS 2006;132:1258-61
2. Argarwal et al. Circ Cardiovasc Interv 2009;2:565-573
3. RogersJH.Circulation2009:119: 2718-25


 July 08, 2024
July 08, 2024