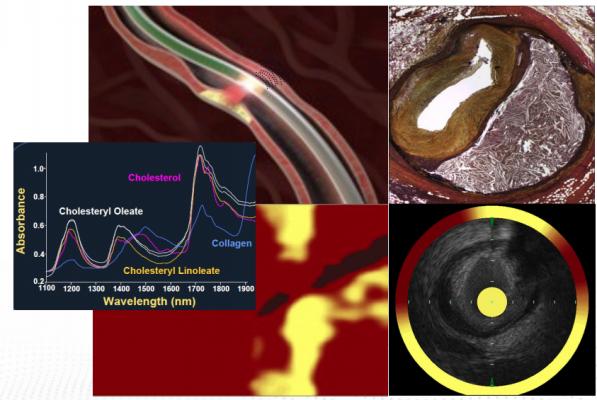
Illustration showing how the near-infrared spectroscopy (NIRS) intravascular ultrasound (IVUS), of NIRS-IVUS, intracoronary imaging system can help identify angiographically non-obstructive lesions with high-risk characteristics for future adverse cardiac outcomes. The system can identify unstable, vulnerable plaques.
October 18, 2020 – New data from the PROSPECT II study shows that near-infrared spectroscopy (NIRS) intravascular ultrasound (IVUS), of NIRS-IVUS, intracoronary imaging can help identify angiographically non-obstructive lesions with high-risk characteristics for future adverse cardiac outcomes.
Findings were reported today at 2020 Transcatheter Cardiovascular Therapeutics (TCT) Connect virtual symposium of the Cardiovascular Research Foundation (CRF).
“In PROSPECT II, lipid-rich plaques, as detected by NIRS, identified angiographically mild non-flow limiting lesions responsible for future coronary events,” said David Erlinge, M.D., Ph.D. Erlinge is professor, Department of Cardiology, clinical sciences, Lund University at Skane University Hospital in Sweden. “High lipid content, along with large plaque burden and small lumen area, may be added as prognostic indices of vulnerable plaques that put patients at risk for adverse outcomes. Further studies prospectively utilizing this information are warranted to improve outcomes for high-risk patients with coronary artery disease.”
Acute coronary syndromes most often arise from rupture and thrombosis of lipid-rich atherosclerotic plaques that develop in the setting of systemic inflammation. In the PROSPECT study, a large plaque burden or small lumen area assessed by IVUS identified angiographically mild lesions at increased risk to cause future coronary events. While lipid-rich fibro-atheromas identified by IVUS can be predictive, plaque interpretation with this technique is not as precise.
Lipid content may be more accurately measured by NIRS. Recent studies have suggested that lipid-rich plaques detected by intracoronary NIRS imaging are associated with adverse outcomes. Identifying these “vulnerable” plaques before they progress may help to inform pharmacologic or other strategies to stabilize the plaque.
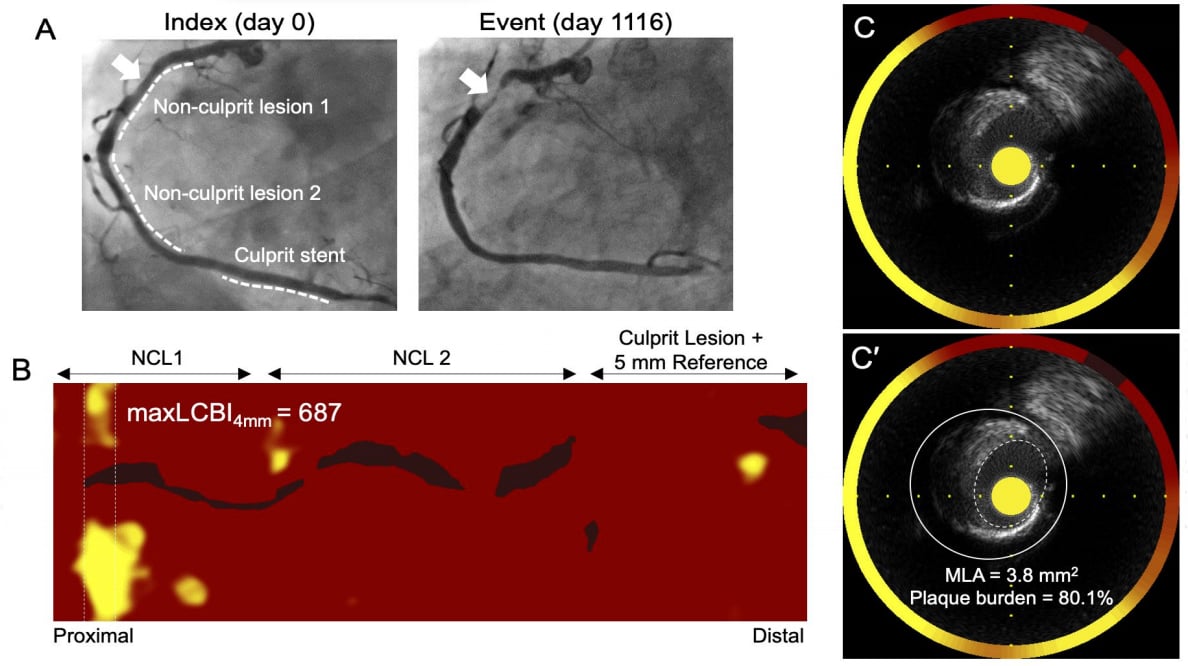
An example if NIRS-IVUS intravascular imaging technology from the PROSPECT II trial showing 3 lesions in coronary artery and the technology chemogram showing areas of high lipid content in the vessel wall in yellow.
PROSPECT II was an investigator-sponsored multicenter, prospective, natural history study conducted at 16 centers in Sweden, Denmark and Norway to validate the use of NIRS and IVUS imaging in identifying non-obstructive lipid-rich plaques that are prone to cause future cardiac events.
After successful treatment of all flow-limiting lesions in patients with recent myocardial infarction, imaging of all three coronary arteries was performed with a combination NIRS-IVUS catheter. The lipid content of “non-culprit” lesions was assessed by NIRS, and IVUS assessment was also performed. The primary outcome was the covariate-adjusted rate of major adverse cardiac events (the composite of cardiac death, myocardial infarction, unstable angina, or progressive angina) adjudicated to have arisen from non-culprit lesions. The relationship between plaques with high lipid content, large plaque burden, and small lumen areas and patient-level and lesion-level events were tested hierarchically.
A total of 3,629 untreated non-culprit lesions were prospectively characterized in 898 patients with myocardial infarction. Adverse events within four years occurred in 13.2% of patients, with a total of 8% of events arising from untreated non-culprit lesions with mean baseline diameter stenosis 46.9%. The presence of highly lipidic lesions was an independent predictor of patient-level non-culprit events (adjusted OR 2.27; 95% CI 1.25-4.13) and lesion-specific events (adjusted OR 7.83; 95% CI 4.12-14.89). Large plaque burden and small lumen areas were also independent predictors of adverse events.
The PROSPECT II trial was funded by research grants from Abbott Vascular, Infraredx and The Medicines Company. Erlinge reported the following disclosures: speaker fees from Amgen, AstraZeneca, Bayer, and Chiesi; and fees for Advisory Boards from Bayer, Boehringer Ingelheim, and Sanofi.
For more information: www.tctconference.com
Related NIRS Content:
VIDEO: Near Infrared Spectroscopy Detects, Aids Risk Assessment of Vulnerable Plaques — Interview with Ron Waksman, M.D.
Find additional TCT 2020 news, video and late-breaking studies


 May 06, 2026
May 06, 2026 









