May 11, 2011 – Every day, Jack Luzovich steps on a special scale that helps keep him in his northern Minnesota home, rather than the hospital. At age 61, he is fighting the debilitating symptoms of congestive heart failure. Last summer, his health hit a low point. “I was retaining fluid and my weight was going up and down,” he remembers. “I couldn’t walk more than 10 to 15 feet. I was on oxygen, jaundiced and had almost no kidney function.”
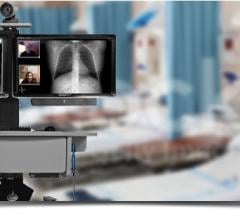
That’s when Jack received a telemonitoring scale from the Essentia Health Heart Failure Program (formerly St. Mary’s Duluth Clinic Heart Failure Program). The device records his weight and asks him questions about his health. It transmits that vital information to a cardiac nurse, who can make changes to his medications and track his condition on a daily basis.
This program, which began more than a decade ago in Duluth, Minn., has posted dramatic results. While national hospital readmission rates for congestive heart failure patients at six months are 40 to 50 percent, Essentia’s sickest heart failure patients, who use the telescale, have readmission rates from 0 to 2 percent. Readmission rates for all patients in the program have hovered around 3 to 7 percent.
The cost benefit is also impressive. An Essentia Health study conducted in partnership with insurance company Blue Cross Blue Shield of Minnesota showed $1.25 million in savings for just 29 patients over six months. These statistics caught the attention of a federal program designed to quickly spread great healthcare ideas across the nation – the Health Care Innovations Exchange, part of the Agency for Healthcare Research and Quality (AHRQ). AHRQ invited Linda Wick, N.P., Heart Failure Program manager, to showcase this concept on Wednesday, May 11, before a panel representing investors, insurers, charitable foundations and healthcare consultants at the AHRQ Innovations Exchange in Rockville, Md.
The group discussed how this program’s success could be replicated at other institutions around the country. “The Innovations Exchange does a rigorous review of programs before deciding to share them nationwide,” Wick says.
Being recognized by the Health Care Innovations Exchange is one of the many ways Essentia Health is providing creative solutions to healthcare challenges, says Chief Medical Officer Carl Heltne, M.D. A cardiologist by training, Heltne founded the Heart Failure Program in 1998.
“Our vision for Essentia Health is to be a national leader in providing high quality, cost-effective integrated healthcare services, particularly in rural areas,” said Heltne. “This program improves quality of life while substantially reducing costs. It’s exactly the type of leadership we are talking about.”
Luzovich sees the value of the program for heart failure patients like him. A year after he began using the scale, his health is greatly improved. He’s back enjoying his favorite hobbies, like woodworking in his basement. “Now I’m running up and down stairs,” he said. “Before, it was up two steps and stop.”
Luzovich had a left ventricular assist device placed last summer, which keeps his heart pumping. As he waits on the heart transplant list, he continues to use his telescale and keeps in regular contact with his caregivers at Essentia Health.
“It’s been a lifesaver for my wife and me,” Luzovich said. “This keeps you from getting into really major trouble. The nurses in the program have become like family.”
For more information: essentiahealth.org


 August 19, 2020
August 19, 2020