Getty Images
May 13, 2024 — Even though mortality and hospitalization rates have improved, the quality of life for those living with hypertrophic cardiomyopathy (HCM) can be compromised with limiting symptoms such as exertional dyspnoea and decreased exercise capacity. A major cause of this in HCM patients is left ventricular outflow tract (LVOT) obstruction, which results in elevated intracardiac pressures. This study demonstrated that aficamten enhanced HCM patients’ exercise capacity with significant improvement in peak oxygen uptake (pVO2), improvement in limiting symptoms, and decreases in LVOT pressure gradients. The late breaking research presented today at Heart Failure 2024, a scientific congress of the European Society of Cardiology (ESC).1
“The SEQUOIA-HCM trial demonstrated that aficamten can reliably and safely eliminate LVOT obstruction in patients with obstructive HCM using a simple and stepwise dosing regimen, and was associated with substantial improvements in clinically relevant endpoints such as exercise capacity and symptoms,” said principal investigator Professor Martin Maron of the Lahey Hospital and Medical Center, Burlington, Massachusetts, US. “HCM patients are often on multiple medications, which frequently provide suboptimal benefit, while aficamten was highly effective at providing clinical improvement as combination therapy, but also as monotherapy.”
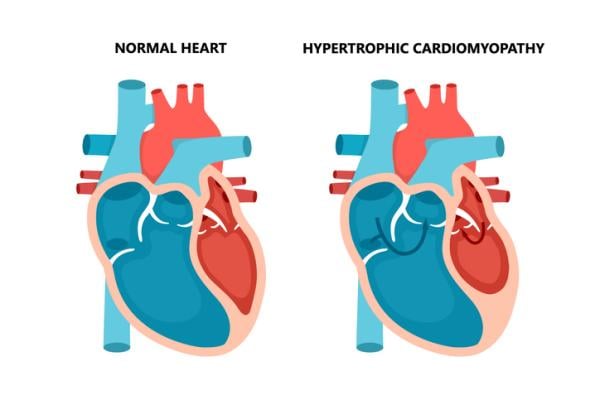
HCM occurs in approximately one in 200 to 500 individuals, with 70% of patients having obstructive disease.2 The condition causes the walls of the left ventricle to become thick and stiff, which can also result in obstruction to blood flow out of the heart and increased intracardiac pressures.
Aficamten is a cardiac myosin inhibitor that was previously shown to reduce LVOT gradients in a phase 2 trial.3 The phase 3 SEQUOIA-HCM trial evaluated the efficacy and safety of aficamten versus placebo in adults with symptomatic obstructive HCM. The primary endpoint was the change in pVO2, assessed using cardiopulmonary exercise testing, from baseline to week 24. Secondary endpoints at 24 weeks included the change in KCCQ score; the proportion of patients with ≥1 class improvement in New York Heart Association (NYHA); change in Valsalva LVOT gradient; the proportion of patients with Valsalva LVOT gradient <30 mmHg; and eligibility for invasive septal reduction.
SEQUOIA-HCM included 282 patients from 101 sites in 14 countries in North America, Asia, and Europe, making it the largest-ever obstructive HCM trial. All participants had reduced exercise capacity due to obstructive HCM. Patients were randomized 1:1 to aficamten or placebo on top of their background medical therapy. The starting dose of aficamten was 5 mg once daily with opportunities at weeks 2, 4, and 6 to increase the dose in 5 mg increments to a maximum dose of 20 mg. Dose adjustments were made according to left ventricular ejection fraction and LVOT gradients assessed using echocardiography.
The mean increase in pVO2 from baseline to 24 weeks was 1.8 ml/kg/min with aficamten compared to 0.0 ml/kg/min with placebo (least-squares mean difference between groups, 1.7 ml/kg/min; 95% confidence interval [CI] 1.0, 2.4; p<0.001). Regarding secondary endpoints at 24 weeks, aficamten resulted in a least-squares mean difference of 7 points in KCCQ score relative to placebo (95% CI 5, 10; p<0.0001). A ≥1 NYHA class improvement was observed in 58.5% of patients on aficamten and 24.3% of patients on placebo (p<0.0001). Aficamten led to a 50 mmHg greater reduction in Valsalva LVOT gradient versus placebo (95% CI -57, -44; p<0.0001). Some 49.3% of patients on aficamten achieved a Valsalva LVOT gradient <30 mmHg versus 3.6% of patients on placebo (p<0.0001). The aficamten group had 78 fewer days eligible for invasive septal reduction compared with the placebo group (p<0.0001).
Professor Maron said: “It was impressive to see that the beneficial effects of aficamten occurred rapidly and consistently over the treatment period and that the doses could be adjusted effectively and safely using only site read echocardiographic measures. It was also reassuring to see that in the very small number of patients found to have an ejection fraction below 50% on aficamten, there was no associated heart failure or the need for dose interruption, and that the effect on ejection fraction was reversible with treatment discontinuation.”
For more information: www.escardio.org
Find more ESC24 conference coverage here
References and notes
1The ‘SEQUOIA-HCM’ trial will be presented during the session ‘Late Breaking Clinical trials: LVAD, HFpEF and hypertrophic cardiomyopathy’ which takes place on 13 May 2024 at 13:45 WEST in Room 1.
2Maron MS, Olivotto I, Zenovich AG, et al. Hypertrophic cardiomyopathy is predominantly a disease of left ventricular outflow tract obstruction. Circulation. 2006;114:2232–2239.
3Maron MS, Masri A, Choudhury L, et al. Phase 2 study of aficamten in patients with obstructive hypertrophic cardiomyopathy. J Am Coll Cardiol. 2023;81:34–45.


 June 08, 2023
June 08, 2023 

