September 9, 2015 — SRI International and DataSpeaks Inc. announced that SRI has licensed the Science of Individuality Measurement Algorithm (SIMA) software from DataSpeaks. SRI will integrate SIMA into the development of a secure enterprise system for clinical decision-making.
The SIMA algorithm can transform multiple streams of patient-generated and sensor data (from wearable or implantable devices) into actionable, integrated, and personalized assessments of risks (harms) and benefits for drugs in a patient's regimen. The resulting scores may be used in the future to quickly alert healthcare practitioners about potential needs for dose adjustments and medication switches.
"The unique SIMA analytic software has the capacity to synergize with other SRI platforms as a component of an integrated enterprise system designed to enhance clinical decision-making, thereby maximizing the likelihood of treatment benefit and minimizing the risk of significant side-effects related to drugs," said David Sahner, M.D., senior director, clinical translation, SRI Biosciences. "This offers the hope of major reductions in morbidity, mortality and the over $7 billion in annual healthcare costs related to the use of potentially inappropriate medications."
The in-licensing of the software was enabled by a grant from the Michigan Economic Development Corporation to establish an inter-institutional Center of Innovation focused on personalized medicine and pharmacotherapy in the elderly.
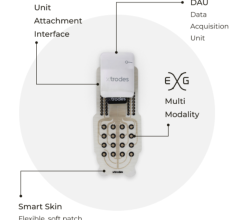
DataSpeaks' proprietary SIMA algorithm applies to multivariate time series data that are becoming ubiquitous with hospital, home, wearable, implanted, and environmental monitoring devices; apps; electronic diaries; and pill monitors. SIMA yields internally standardized scores that quantify evidence for clinically relevant interactions over time as "edges" in network graphs that are computed from data about individual patients.
Additional potential uses of SIMA include improvement of diagnostic taxonomies that may facilitate better prediction and therapy choices, identification of disease mechanisms and new drug development targets, and use in novel clinical trial designs that do not "wash out" individual patient differences.
For more information: www.dataspeaks.com, www.sri.com


 April 06, 2026
April 06, 2026