October 30, 2007 - Cordis Corp. will launch in the U.S. and European its next generation carotid stent system, the PRECISEPRO RX Nitinol Self-Expanding Stent, to treat clogged neck arteries, in order to offer physicians a broader range of treatment options for patients with carotid artery disease.
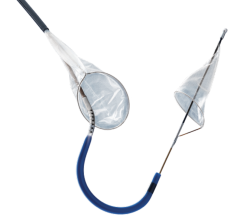
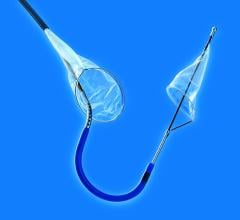
With reportedly the lowest profile system approved in the U.S., the PRECISE Stent is a small, wire-mesh tube that props open the blocked artery and may be used in conjunction with Cordis’ ANGIOGUARDRx Emboli Capture Guidewire System - a tiny, basket-like device designed to trap particles of plaque, blood clot or other material that may be dislodged in the carotid artery during stent placement.
The lower profile carotid stent system is designed to make it easier to use and enable improved crossing of the lesion. Plus, the delivery system is said to enable more efficient manipulation of the catheter and guidewire during stenting procedures.
For more information: www.cordis.com


 April 25, 2023
April 25, 2023