July 20, 2022 — Teleflex Incorporated, a leading global provider of medical technologies, announced the first patient enrollment in a clinical study that is intended to demonstrate the safety of MANTA Vascular Closure Device ultrasound (U/S) guided closure, without dependence on the pre-procedural depth locator measurements. The study will enroll patients undergoing elective TAVR procedures with planned percutaneous femoral arterial access.
Enrolling up to 150 patients in up to 15 investigational sites across the US and Canada, the MANTA Ultra Study is a prospective, multi-center, single-arm trial employing the primary safety endpoint of any large bore access-site related Valve Academic Research Consortium-2 (VARC-2) Major Vascular Complication within 30 days (adapted from VARC-2 Criteria).The occurrence of access site complications is known to be associated with higher rates of morbidity and mortality1 and increased costs associated with prolonged length of stay.2 The practice of ultrasound guided access has been associated with a reduced risk of major and minor access site vascular complications, as well as reduced risk of life threatening, major, and minor access site bleed complications.3 Teleflex is committed to exploring whether the known benefits of ultrasound-guided access will translate to ultrasound-guided closure, resulting in safe MANTA Device deployment.
“The resourcing of the MANTA Ultra Study by Teleflex reflects our commitment to providing interventional cardiologists and surgeons with category-defining tools that enable difficult procedures and minimize complications – in this case, the complications attributable to large bore access that are inherent to TAVI procedures,” said Teleflex Medical Director, Chris Buller, MD.
The principal investigators of the study include David A. Wood*, MD, Head, VGH Division of Cardiology and Director, University of British Columbia Centre for Cardiovascular Innovation, Vancouver, British Columbia, and Vijay S. Iyer*, MD, Chief, Cardiovascular Medicine and Associate Professor of Medicine at the University of Buffalo, Buffalo, New York.
“We’re excited to embark on the Teleflex MANTA Ultra study,” said Dr. Wood. “We expect the study results to show that the use of ultrasound further aids in avoiding MANTA Device deployment complications attributed to anchor mal-positioning by providing direct visualization of the closure process.”
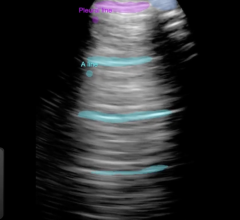
With visualization of the access site during MANTA Device deployment, the study intends to demonstrate that ultrasound allows for precise deployment and positive positioning of the MANTA Device anchor within the vessel. This approach, if proven effective, will allow the MANTA Device to be used in clinical scenarios where the baseline arterial depth is not measured, or may have changed intra-operatively or post-operatively. MANTA Device ultrasound guided closure is intended to be an alternative method to the conventional pre-procedural depth locator measurement technique that can provide additional procedural flexibility to operators.
In addition to the primary safety endpoint, the study will employ a secondary endpoint of any large bore access-site related VARC-2 minor vascular complication within 30 days (adapted from VARC-2 Criteria) and secondary effectiveness endpoints that include time to hemostasis, technical success, ambulation success, time to ambulation, treatment success and procedure time – all with 30-day and 12-month follow-up time points.
For more information: teleflex.com


 January 28, 2026
January 28, 2026