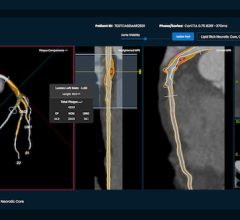
December 11, 2007 - GE’s SnapShot Pulse technology for prospectively gated diagnostic cardiac CT exams has been proven to reduce a patient’s radiation exposure by up to 83 percent and improve image quality, validated from clinicians performing thousands of exams at sites around the world.
Also reducing exposure is the VolumeShuttle, delivering twice the anatomical coverage (80 millimeters) with up to 24 percent less radiation exposure compared to a conventional 40-millimeter cine perfusion protocol. This single injection acquisition generates clinically comparable perfusion maps and angiographic studies and allows clinicians to see more anatomy, enabling whole organ anatomical and physiological assessment.
Both SnapShot Pulse and VolumeShuttle are offered on the GE LightSpeed VCT XT configuration, enabled by real-time scan control architecture.
For more information: www.gehealthcare.com


 April 15, 2026
April 15, 2026