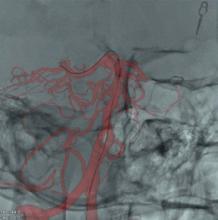
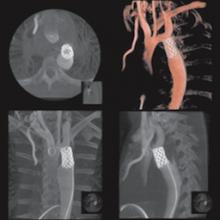
As catheter-based, minimally invasive procedures expand rapidly beyond treatment of the coronary arteries into all areas of the human anatomy, angiography X-ray imaging systems have moved beyond their original purpose of simple vascular imaging. Today, in addition to interventional cardiology, cath labs are frequently shared by numerous specialists, including interventional radiology, interventional oncology, electrophysiology (EP), vascular surgeons and neurology.
If you enjoy this content, please share it with a colleague
- Read more about Angiography No Longer Limited to Cardiology
- Log in or register to post comments