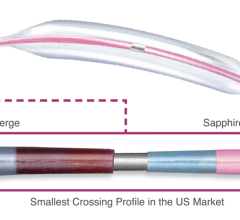
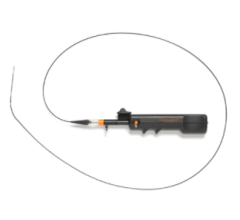
March 3, 2016 — Avinger Inc. announced the company has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for an enhanced version of its Pantheris lumivascular atherectomy system for the treatment of peripheral artery disease (PAD). Avinger will commence U.S. commercialization of this enhanced version of Pantheris immediately, which features improved ergonomics, physician controls and manufacturability
PAD affects nearly 20 million adults in the United States and over 200 million people globally. It is caused by a buildup of plaque in the arteries that blocks blood flow to the legs and feet. Often dismissed as normal signs of aging, symptoms of PAD include painful cramping, numbness, or discoloration in the legs or feet. PAD can become so severe and difficult to address with traditional treatments that patients and physicians often resort to invasive bypass surgeries, which can result in even greater health risks and lengthy, painful recoveries. In severe cases, patients often face amputation, the worst-case scenario associated with PAD.
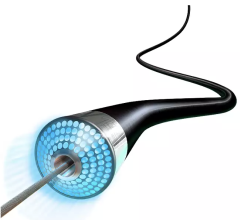
Atherectomy is a minimally invasive treatment for PAD in which a catheter-based device is used to remove plaque from a blood vessel. Lumivascular technology utilized in the Pantheris system allows physicians to see from inside the artery during a directional atherectomy procedure using an imaging modality called optical coherence tomography (OCT). In the past, physicians have had to rely solely on X-ray as well as touch and feel to guide their tools while they try to treat complicated arterial disease. With the lumivascular approach, physicians can more accurately navigate their devices and treat PAD lesions, thanks to the OCT images they see from inside the artery.
“Atherectomy is a proven treatment that relieves pain and restores blood flow, and Pantheris has been eagerly anticipated in the clinical community because it is a leap forward in atherectomy technology compared to what we have had in the past,” said Thomas Davis, M.D., of St. John Hospital and Medical Center in St. Clair Shores, Mich. “Now, for the first time, we are able to see exactly where we are removing the plaque, and are better able to leave the healthy artery alone. Clinical data suggests that the precision offered with Pantheris will have a lasting positive effect on patient outcomes. In addition, Pantheris visualizes the artery without requiring ionizing radiation. Thus, OCT has the potential to reduce physician and patient X-ray exposure, which is a concern for all clinicians performing multiple procedures.”
Pantheris has demonstrated clinical effectiveness and an excellent safety profile. In the recently completed VISION study, 130 patients were treated with Pantheris and followed for a period of six months. The study achieved its primary safety and efficacy endpoints and showed a target lesion revascularization rate of just 8 percent, and not a single event of vessel perforation, clinically significant dissection or late aneurysm resulted from Pantheris.
In October 2015, Avinger received 510(k) clearance from the FDA for the Pantheris system for the treatment of patients with PAD. The company subsequently filed a second 510(k) application in December 2015 seeking clearance for an enhanced version of the system, including improved ergonomics, physician controls and manufacturability.
For more information: www.avinger.com


 November 08, 2024
November 08, 2024