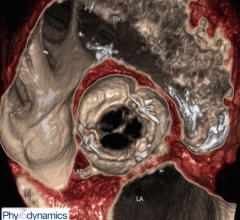
Fysicon announced that it has been granted 510(k) clearance by the U.S. Food and Drug…
Hitachi Healthcare Americas Inc. announced it has attained U.S. Food and Drug Administration (…
September 15, 2017 — Philips highlighted its newest solution for…
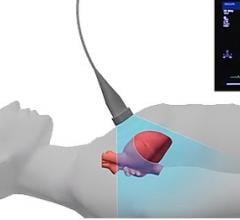
September 7, 2017 — Sapheneia and Scannerside received U.S.
The U.S. Food and Drug Administration (FDA) has cleared TrueFusion, a new cardiovascular…
Abbott announced it has received U.S. Food and Drug Administration (FDA) approval for its Full…
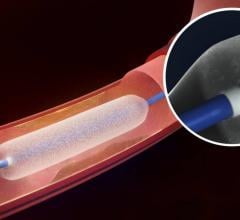
C.R. Bard Inc. announced the Lutonix 035 Drug Coated Balloon PTA Catheter (DCB) has been granted…
Biotronik announced U.S. Food and Drug Administration (FDA) approval and commercial availability…
Aug.
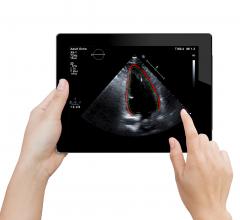
August 10, 2017 — CDN recently announced a new partnership agreement with DiA Imaging Analysis…
AUM Cardiovascular announced it has received clearance from the U.S. Food and Drug…
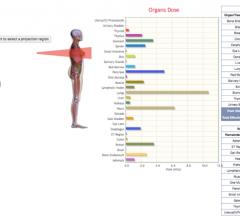
Virtual Phantoms Inc. announced the release of VirtualDoseIR, a tool for assessing organ dose…
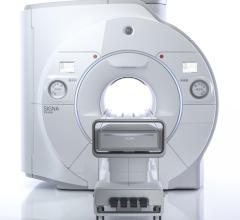
GE Healthcare announced Signa Premier, a new wide bore 3.0T magnetic resonance imaging (MRI)…
The Spectranetics Corp. announced receipt of U.S. Food and Drug Administration (FDA) pre-market…
TomTec Zero is the latest addition to the TomTec portfolio. Now every physician reading…
Samsung announced U.S. Food and Drug Administration (FDA) approval of the BodyTom Elite, an…
Cardiologs Technologies SAS announced that it has received U.S. Food and Drug Administration (…
July 12, 2017 — At the 2017 annual meeting for the Society of Cardiovascular Computed Tomography…
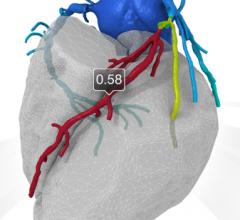
July 6, 2017 — GE Healthcare and HeartFlow Inc.
June 27, 2017 — Toshiba Medical, a Canon Group company, introduced its newest…

 September 18, 2017
September 18, 2017